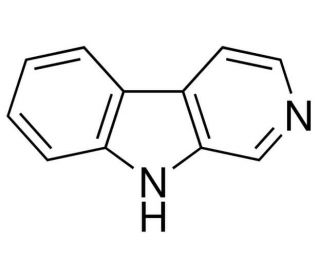
Norharmane (CAS 244-63-3)
See product citations (33)
QUICK LINKS
Norharmane, also known as β-carboline, is a compound that is investigated in the context of analytical chemistry and environmental studies. In analytical applications, norharmane is frequently used as a matrix substance in matrix-assisted laser desorption/ionization (MALDI) mass spectrometry due to its strong ultraviolet absorption and ability to assist in the ionization of analytes without fragmenting them. This is particularly useful in the study of large biomolecules like proteins, peptides, and nucleic acids. Furthermore, norharmane is also of interest in the study of photochemical processes, where its photophysical properties are valuable for understanding the behavior of aromatic compounds upon exposure to light. Researchers also use norharmane to explore its presence in various environmental samples, such as tobacco smoke and grilled foods, due to its formation during the pyrolysis of tryptophan. In addition, norharmane is a subject in the study of intermolecular interactions and self-assembly processes, providing insights into the fundamental principles governing molecular recognition and complex formation.
Norharmane (CAS 244-63-3) References
- Photochemistry of norharmane in aqueous solution. | Gonzalez, MM., et al. 2009. Photochem Photobiol Sci. 8: 1139-49. PMID: 19639116
- Effect of norharmane in vitro on juvenile hormone epoxide hydrolase activity in the lower termite, Reticulitermes speratus. | Itakura, S., et al. 2008. J Insect Sci. 8: 13. PMID: 20345286
- Photosensitized electron transfer within a self-assembled norharmane-2'-deoxyadenosine 5'-monophosphate (dAMP) complex. | Gonzalez, MM., et al. 2012. Org Biomol Chem. 10: 9359-72. PMID: 23111419
- Effects of adolescent treatment with nicotine, harmane, or norharmane in male Sprague-Dawley rats. | Goodwin, AK., et al. 2015. Neurotoxicol Teratol. 47: 25-35. PMID: 25450662
- Norharmane rhenium(I) polypyridyl complexes: synthesis, structural and spectroscopic characterization. | Maisuls, I., et al. 2015. Dalton Trans. 44: 17064-74. PMID: 26365709
- Screening of Norharmane from Seven Cyanobacteria by High-performance Liquid Chromatography. | Karan, T. and Erenler, R. 2017. Pharmacogn Mag. 13: S723-S725. PMID: 29142439
- Antibiofilm activities of norharmane and its derivatives against Escherichia coli O157:H7 and other bacteria. | Lee, JH., et al. 2017. Phytomedicine. 36: 254-261. PMID: 29157822
- Cross state-dependent memory retrieval between morphine and norharmane in the mouse dorsal hippocampus. | Ebrahimi-Ghiri, M., et al. 2019. Brain Res Bull. 153: 24-29. PMID: 31400494
- A natural indole alkaloid, norharmane, affects PIN expression patterns and compromises root growth in Arabidopsis thaliana. | López-González, D., et al. 2020. Plant Physiol Biochem. 151: 378-390. PMID: 32278957
- Streamlined Analysis of Cardiolipins in Prokaryotic and Eukaryotic Samples Using a Norharmane Matrix by MALDI-MSI. | Yang, H., et al. 2020. J Am Soc Mass Spectrom. 31: 2495-2502. PMID: 32924474
- Phytotoxic Activity of the Natural Compound Norharmane on Crops, Weeds and Model Plants. | López-González, D., et al. 2020. Plants (Basel). 9: PMID: 33050191
- Inhibitory effect of norharmane on Serratia marcescens NJ01 quorum sensing-mediated virulence factors and biofilm formation. | Luo, HZ., et al. 2021. Biofouling. 37: 145-160. PMID: 33682541
- Nitrovinylindoles as Heterotrienes: Electrocyclic Cyclization En Route to β-Carbolines: Total Synthesis of Alkaloids Norharmane, Harmane, and Eudistomin N. | Aksenov, NA., et al. 2022. Org Lett. 24: 7062-7066. PMID: 36166488
- Effects of metyrapone and norharmane on microsomal mono-oxygenase and epoxide hydrolase activities. | Bulleid, NJ. and Craft, JA. 1984. Biochem Pharmacol. 33: 1451-7. PMID: 6732863
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Norharmane, 100 mg | sc-212410 | 100 mg | $55.00 | |||
Norharmane, 500 mg | sc-212410A | 500 mg | $148.00 | |||
Norharmane, 1 g | sc-212410C | 1 g | $245.00 | |||
Norharmane, 5 g | sc-212410D | 5 g | $408.00 | |||
Norharmane, 25 g | sc-212410E | 25 g | $1642.00 | |||
Norharmane, 50 g | sc-212410F | 50 g | $3256.00 |
