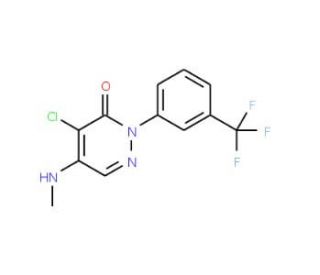
Molecular structure of Norflurazon, CAS Number: 27314-13-2
Norflurazon (CAS 27314-13-2)
Alternate Names:
4-Chloro-5-(methylamino)-2-(α,α,α-trifluoro-m-tolyl)-3(2H)-pyridazinone
Application:
Norflurazon is a selective herbicide
CAS Number:
27314-13-2
Molecular Weight:
303.67
Molecular Formula:
C12H9ClF3N3O
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Norflurazon is a common selective herbicide used to control the proliferation of broadleaf weeds and seasonal grasses. Norflurazon is a herbicide that prevents the growth of weeds by impeding the production of carotenoids. It is a pyridazinone with a pyridazin-3(2H)-one structure that has m-(trifluoromethyl)phenyl, chloro, and methylamino groups at positions 2, 4, and 5, respectively. It is utilized as a pre-emergence weed killer for both grasses and broad-leafed weeds in various crops. Norflurazon has the appearance of colorless and odorless crystals and is not corrosive.
Norflurazon (CAS 27314-13-2) References
- Preparation and characterization of inclusion complex of norflurazon and beta-cyclodextrin to improve herbicide formulations. | Villaverde, J., et al. 2004. J Agric Food Chem. 52: 864-9. PMID: 14969543
- Effect of soil type on adsorption-desorption, mobility, and activity of the herbicide norflurazon. | Morillo, E., et al. 2004. J Agric Food Chem. 52: 884-90. PMID: 14969546
- Fluridone and norflurazon, carotenoid-biosynthesis inhibitors, promote seed conditioning and germination of the holoparasite Orobanche minor. | Chae, SH., et al. 2004. Physiol Plant. 120: 328-337. PMID: 15032868
- Toxicity testing of herbicide norflurazon on an aquatic bioindicator species--the planarian Polycelis felina (Daly.). | Horvat, T., et al. 2005. Aquat Toxicol. 73: 342-52. PMID: 15899527
- Norflurazon mobility, dissipation, activity, and persistence in a sandy soil as influenced by formulation. | Sopeña, F., et al. 2007. J Agric Food Chem. 55: 3561-7. PMID: 17407320
- Effect of norflurazon on resorcinolic lipid metabolism in rye seedlings. | Magnucka, EG., et al. 2007. Z Naturforsch C J Biosci. 62: 239-45. PMID: 17542491
- Evidence that norflurazon affects chloroplast lipid unsaturation in soybean leaves (Glycine max L.). | Abrous-Belbachir, O., et al. 2009. J Agric Food Chem. 57: 11434-40. PMID: 19951003
- Norflurazon and simazine removal from surface water using a constructed wetland. | Wilson, PC., et al. 2011. Bull Environ Contam Toxicol. 87: 426-30. PMID: 21833732
- Photostabilization of the herbicide norflurazon microencapsulated with ethylcellulose in the soil-water system. | Sopeña, F., et al. 2011. J Hazard Mater. 195: 298-305. PMID: 21924830
- Sonophotocatalytic mineralization of Norflurazon in aqueous environment. | Sathishkumar, P., et al. 2016. Chemosphere. 146: 216-25. PMID: 26735720
- Determination of norflurazon concentration in wheat leaves using a modified QuEChERS method. | Trzebuniak, KF. and Mysliwa-Kurdziel, B. 2017. Acta Biochim Pol. 64: 431-436. PMID: 28822995
- genome uncoupled1 Mutants Are Hypersensitive to Norflurazon and Lincomycin. | Zhao, X., et al. 2018. Plant Physiol. 178: 960-964. PMID: 30154176
- At3g53630 encodes a GUN1-interacting protein under norflurazon treatment. | Huang, XQ., et al. 2021. Protoplasma. 258: 371-378. PMID: 33108535
- Alleviation of norflurazon-induced photobleaching by overexpression of Fe-chelatase in transgenic rice. | Park, JH. and Jung, S. 2021. J Pestic Sci. 46: 258-266. PMID: 34566459
Inhibitor of:
PhyB.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Norflurazon, 100 mg | sc-250580 | 100 mg | $188.00 |
