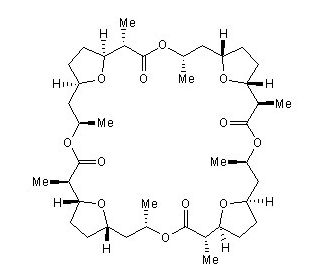
Nonactin (CAS 6833-84-7)
See product citations (1)
QUICK LINKS
Nonactin is a cyclic ionophore and part of the macrotetrolide antibiotics family. The ionophore is produced by several species of Streptomyces which selectively binds K+ and NH4+. Nonactin has been recorded to function by binding a metal ionophore and transporting it across cell membranes for release. The compound can functionally perform this transport due to its conformational flexibility and hydrophobic exterior. The compound forms a natural diastereomer which is required for its antibacterial and binding capacities. At a concentration of 1x10-7 M Nonactin uncoupled oxidative phosphorylation and induced ATP hydrolysis. The compound has been observed to specifically inhibit the processing of cytoplasmic precursor proteins destined for the mitochondria. In the presence of Thallus acetate, Nonactin has further stimulated the swelling of rat liver mitochondria than thallus acetate alone. It also has inhibited the effects on P170 glycoprotein-mediated efflux of chemotherapeutic agents in MDR cancer cells. Nonactin may act to inhibit the production and gene transcription of IL-5 but not of IL-2 or IL-4. Nonactin has also been used as an ammonium ionophore for ammonium ion determination in a potentiometric electrode for biosensor construction as well as in a fluorescent immuno assay to detect K+ ions in pharmaceutical samples.
Nonactin (CAS 6833-84-7) References
- Nonactin biosynthesis: the product of nonS catalyzes the formation of the furan ring of nonactic acid. | Woo, AJ., et al. 1999. Antimicrob Agents Chemother. 43: 1662-8. PMID: 10390219
- Determination of potassium ions in pharmaceutical samples by FIA using a potentiometric electrode based on ionophore nonactin occluded in EVA membrane. | Garcia, CA., et al. 2003. J Pharm Biomed Anal. 31: 11-8. PMID: 12560044
- A new potentiometric ammonium electrode for biosensor construction. | Karakuş, E., et al. 2006. Artif Cells Blood Substit Immobil Biotechnol. 34: 523-34. PMID: 16893815
- Nonactin biosynthesis: setting limits on what can be achieved with precursor-directed biosynthesis. | Kusche, BR., et al. 2009. Bioorg Med Chem Lett. 19: 1233-5. PMID: 19167217
- Alternating pattern of stereochemistry in the nonactin macrocycle is required for antibacterial activity and efficient ion binding. | Kusche, BR., et al. 2009. J Am Chem Soc. 131: 17155-65. PMID: 19902940
- Efficient production of nonactin by Streptomyces griseus subsp. griseus. | Zhan, Y. and Zheng, S. 2016. Can J Microbiol. 62: 711-4. PMID: 27405846
- On the ionophoric selectivity of nonactin and related macrotetrolide derivatives. | Martínez-Haya, B., et al. 2017. Phys Chem Chem Phys. 19: 1288-1297. PMID: 27966685
- Preferential host-guest coordination of nonactin with ammonium and hydroxylammonium. | Martínez-Haya, B., et al. 2018. J Chem Phys. 149: 225101. PMID: 30553267
- Inclusion complexes of the macrocycle nonactin with benchmark protonated amines: aniline and serine. | Avilés-Moreno, JR., et al. 2022. Phys Chem Chem Phys. 24: 8422-8431. PMID: 35343526
- Beyond Nonactin: Potentiometric Ammonium Ion Sensing Based on Ion-selective Membrane-free Prussian Blue Analogue Transducers. | Xu, L., et al. 2022. Anal Chem. 94: 10487-10496. PMID: 35839308
- Antibiotics as tools for metabolic studies. V. Effect of nonactin, monactin, dinactin, and trinactin on oxidative phosphorylation and adenosine triphosphatase induction. | Graven, SN., et al. 1966. Biochemistry. 5: 1729-35. PMID: 4225380
- Antibiotics as tools for metabolic studies. VI. Damped oscillatory swelling of mitochondria induced by nonactin, monactin, dinactin, and trinactin. | Graven, SN., et al. 1966. Biochemistry. 5: 1735-42. PMID: 5962273
- Uptake of thallous ions by mitochondria is stimulated by nonactin but not by respiration alone. | Skulskii, IA., et al. 1981. Eur J Biochem. 120: 263-6. PMID: 6895621
- Mobile ionophores are a novel class of P-glycoprotein inhibitors. The effects of ionophores on 4'-O-tetrahydropyranyl-adriamycin incorporation in K562 drug-resistant cells. | Borrel, MN., et al. 1994. Eur J Biochem. 223: 125-33. PMID: 7518390
- Simple understanding and optimistic strategy for coping with atopic diseases. IL-5 central hypothesis on eosinophilic inflammation. | Okudaira, H. and Mori, A. 1998. Int Arch Allergy Immunol. 117: 11-9. PMID: 9751843
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Nonactin, 5 mg | sc-203164 | 5 mg | $66.00 | |||
Nonactin, 25 mg | sc-203164A | 25 mg | $255.00 |
