Nitrobenzene (CAS 98-95-3)
QUICK LINKS
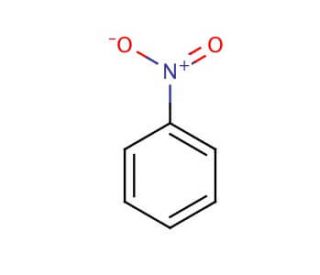
Nitrobenzene is an organic compound with the chemical formula C6H5NO2, characterized by a benzene ring where one hydrogen atom is substituted by a nitro group (-NO2). This pale yellow oily liquid is known for its distinctive almond-like odor and is primarily manufactured through the nitration of benzene using a mixture of concentrated nitric acid and sulfuric acid. The presence of the nitro group significantly impacts the chemical properties of nitrobenzene, making it highly electrophilic and thus a key intermediate in the synthesis of aniline, a precursor to numerous synthetic dyes, pharmaceuticals, and plastics. In research, nitrobenzene has been extensively used to study electrophilic aromatic substitution reactions due to its reactivity, which allows exploration into the kinetics and mechanisms of these reactions under various conditions. The compound also serves as a solvent or a reagent in the synthesis of other complex organic compounds. Its electron-withdrawing nitro group enhances the electrophilic character of the benzene ring, facilitating further nitration or other substitution reactions that are crucial in organic synthesis. Additionally, nitrobenzene has been employed in electrochemical studies where its reduction and oxidation potentials are of interest. In materials science, it has been used in the development and testing of organic semiconductors and other electronic materials, providing insights into charge transfer processes and molecular alignment within these systems.
Nitrobenzene (CAS 98-95-3) References
- [A study on mechanism for cytotoxicity of nitrobenzene to hepatocarcinoma cell line]. | Han, C., et al. 2001. Zhonghua Yu Fang Yi Xue Za Zhi. 35: 48-50. PMID: 11860961
- [Study on removal of nitrobenzene in water by O3/H2O2]. | Ma, J. and Shi, F. 2002. Huan Jing Ke Xue. 23: 67-71. PMID: 12533929
- Chromosomal genotoxicity of nitrobenzene and benzonitrile. | Bonacker, D., et al. 2004. Arch Toxicol. 78: 49-57. PMID: 14513206
- Oxidative Pathway for the Biodegradation of Nitrobenzene by Comamonas sp. Strain JS765. | Nishino, SF. and Spain, JC. 1995. Appl Environ Microbiol. 61: 2308-13. PMID: 16535050
- Nitrobenzene removal in bioelectrochemical systems. | Mu, Y., et al. 2009. Environ Sci Technol. 43: 8690-5. PMID: 20028072
- Changes in Sertoli cell function in vitro induced by nitrobenzene. | Allenby, G., et al. 1990. Fundam Appl Toxicol. 14: 364-75. PMID: 2108073
- Thermal hazard assessment of nitrobenzene/dinitrobenzene mixtures. | Badeen, C., et al. 2011. J Hazard Mater. 188: 52-7. PMID: 21316854
- Enhanced Nitrobenzene reduction by zero valent iron pretreated with H2O2/HCl. | Yang, Z., et al. 2018. Chemosphere. 197: 494-501. PMID: 29407811
- A flow cytometric analysis of cytotoxic effects of nitrobenzene on rat spermatogenesis. | Iida, S., et al. 1997. J Toxicol Sci. 22: 397-407. PMID: 9442449
- High adsorption capacity of nitrobenzene from aqueous solution using activated carbons prepared from vegetable waste | Kecira, Z., Benturki, O., Benturki, A., Daoud, M., & Girods, P. 2020. Environmental Progress & Sustainable Energy. 39(6): e13463.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Nitrobenzene, 25 ml | sc-250568 | 25 ml | $41.00 | |||
Nitrobenzene, 500 ml | sc-250568A | 500 ml | $116.00 |
