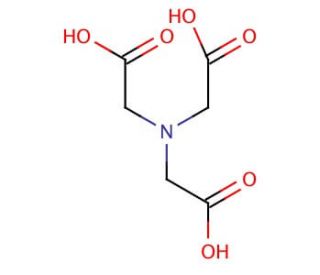
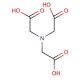
Nitrilotriacetic acid (CAS 139-13-9)
QUICK LINKS
Nitrilotriacetic acid forms stable complexes with metal ions, particularly with transition metals such as iron and copper. It functions by binding to metal ions through its multiple carboxylic acid groups, forming a stable coordination complex. This allows it to effectively sequester metal ions in solution, preventing them from participating in unwanted side reactions or interfering with experimental processes.Nitrilotriacetic acid can also act as a buffer, helping to maintain the pH of a solution within a specific range. Its mechanism of action involves the coordination of metal ions through the formation of coordinate covalent bonds, effectively removing them from the solution and preventing their participation in undesired chemical reactions. This property may be useful in various experimental applications where the presence of metal ions needs to be controlled or minimized.
Nitrilotriacetic acid (CAS 139-13-9) References
- Biodegradation of nitrilotriacetic acid (NTA) and ferric-NTA complex by aerobic microbial granules. | Nancharaiah, YV., et al. 2006. Water Res. 40: 1539-46. PMID: 16600324
- Nitrilotriacetic acid degradation under microbial fuel cell environment. | Jang, JK., et al. 2006. Biotechnol Bioeng. 95: 772-4. PMID: 16958138
- Characterization of the genotoxicity of nitrilotriacetic acid. | Nesslany, F., et al. 2008. Environ Mol Mutagen. 49: 439-52. PMID: 18449932
- Multivalent and flexible PEG-nitrilotriacetic acid derivatives for non-covalent protein pegylation. | Mero, A., et al. 2011. Pharm Res. 28: 2412-21. PMID: 21611874
- Nitrilotriacetic acid: a novel reducing agent for synthesizing colloidal gold. | Njagi, JI. and Goia, DV. 2014. J Colloid Interface Sci. 421: 27-32. PMID: 24594028
- Diarmed (adamantyl/alkyl) surfactants from nitrilotriacetic acid. | Trillo, JV., et al. 2014. Colloids Surf B Biointerfaces. 123: 974-80. PMID: 25465758
- Comparison of Nitrilotriacetic Acid and [S,S]-Ethylenediamine-N,N'-disuccinic Acid in UV-Fenton for the Treatment of Oil Sands Process-Affected Water at Natural pH. | Zhang, Y., et al. 2016. Environ Sci Technol. 50: 10535-10544. PMID: 27588553
- UV/Nitrilotriacetic Acid Process as a Novel Strategy for Efficient Photoreductive Degradation of Perfluorooctanesulfonate. | Sun, Z., et al. 2018. Environ Sci Technol. 52: 2953-2962. PMID: 29397704
- Influence of alkyl polyglucoside, citric acid, and nitrilotriacetic acid on phytoremediation in pyrene-Pb co-contaminated soils. | Liu, X., et al. 2018. Int J Phytoremediation. 20: 1055-1061. PMID: 30095307
- Self-assembling nitrilotriacetic acid nanofibers for tracking and enriching His-tagged proteins in living cells. | Zhang, LS., et al. 2021. J Mater Chem B. 9: 80-84. PMID: 33313613
- Nitrilotriacetic Acid (NTA) and Phenylboronic Acid (PBA) Functionalized Nanogels for Efficient Encapsulation and Controlled Release of Insulin. | Li, C., et al. 2018. ACS Biomater Sci Eng. 4: 2007-2017. PMID: 33445271
- The changes of rhizosphere characteristics contributed to enhanced Pb accumulation in Athyrium wardii (Hook.) Makino after nitrilotriacetic acid application. | Zhang, Y., et al. 2022. Environ Sci Pollut Res Int. 29: 6184-6193. PMID: 34436720
- Evaluation of the Effects of Nitrilotriacetic Acid as a Chelating Agent on the Biochemical Toxicity of Lead in Oreochromis niloticus. | Aytekin, T. 2022. Biol Trace Elem Res. 200: 2908-2914. PMID: 34677762
- Effect of nitrilotriacetic acid and tea saponin on the phytoremediation of Ni by Sudan grass (Sorghum sudanense (Piper) Stapf.) in Ni-pyrene contaminated soil. | Jiao, A., et al. 2022. Chemosphere. 294: 133654. PMID: 35066084
- Simultaneous preconcentration and pre-column derivatization for rapid analysis of nitrilotriacetic acid in environmental waters by high performance liquid chromatography. | Cheng, J., et al. 2022. J Chromatogr A. 1674: 463137. PMID: 35588592
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Nitrilotriacetic acid, 5 g | sc-215578 | 5 g | $31.00 | |||
Nitrilotriacetic acid, 100 g | sc-215578A | 100 g | $72.00 |
