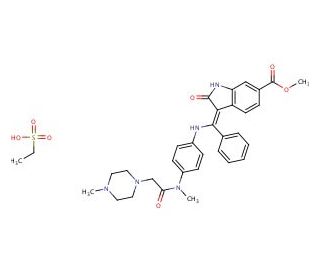
Nintedanib esylate (CAS 656247-18-6)
QUICK LINKS
Nintedanib Esylate is often utilized in research focused on fibrotic processes and the mechanisms involved in fibrosis. It is particularly of interest in studies related to its inhibitory action on multiple tyrosine kinases, which play a role in the signaling pathways that lead to fibrotic diseases. Researchers also investigate Nintedanib Esylate in relation to angiogenesis and the proliferation of vascular endothelial cells. The compound′s mechanism involves blocking the receptors for platelet-derived growth factor (PDGF), fibroblast growth factor (FGF), and vascular endothelial growth factor (VEGF), which are key molecules in these pathological processes. Additionally, Nintedanib Esylate is studied in the context of tissue remodeling, with a focus on understanding how inhibiting these signaling pathways can impact the extracellular matrix and cellular proliferation.
Nintedanib esylate (CAS 656247-18-6) References
- Enhanced bioavailability and intestinal uptake of Gemcitabine HCl loaded PLGA nanoparticles after oral delivery. | Joshi, G., et al. 2014. Eur J Pharm Sci. 60: 80-9. PMID: 24810394
- Anlotinib inhibits angiogenesis via suppressing the activation of VEGFR2, PDGFRβ and FGFR1. | Lin, B., et al. 2018. Gene. 654: 77-86. PMID: 29454091
- Treatment of idiopathic pulmonary fibrosis with Nintedanib: an update. | Wongkarnjana, A., et al. 2019. Expert Rev Respir Med. 13: 1139-1146. PMID: 31564185
- Statistical optimization and validation of a novel ultra-performance liquid chromatography method for estimation of nintedanib in rat and human plasma. | Shukla, SK., et al. 2020. Bioanalysis. 12: 159-174. PMID: 32052636
- Inhibition of colorectal cancer-associated fibroblasts by lipid nanocapsules loaded with acriflavine or paclitaxel. | Fourniols, T., et al. 2020. Int J Pharm. 584: 119337. PMID: 32371002
- Inhalation: A means to explore and optimize nintedanib's pharmacokinetic/pharmacodynamic relationship. | Epstein-Shochet, G., et al. 2020. Pulm Pharmacol Ther. 63: 101933. PMID: 32750409
- Inhaled nintedanib is well-tolerated and delivers key pharmacokinetic parameters required to treat bleomycin-induced pulmonary fibrosis. | Surber, MW., et al. 2020. Pulm Pharmacol Ther. 63: 101938. PMID: 32841676
- Design, synthesis and antitumor evaluation of new 1,8-naphthalimide derivatives targeting nuclear DNA. | Liang, GB., et al. 2021. Eur J Med Chem. 210: 112951. PMID: 33109400
- Development and pharmacokinetic evaluation of a self-nanoemulsifying drug delivery system for the oral delivery of cannabidiol. | Kok, LY., et al. 2022. Eur J Pharm Sci. 168: 106058. PMID: 34763088
- A scalable 3D tissue culture pipeline to enable functional therapeutic screening for pulmonary fibrosis. | Cummins, KA., et al. 2021. APL Bioeng. 5: 046102. PMID: 34805716
- Bioavailability enhancement of vitamin E TPGS liposomes of nintedanib esylate: formulation optimization, cytotoxicity and pharmacokinetic studies. | Kala, SG. and Chinni, S. 2022. Drug Deliv Transl Res. 12: 2856-2864. PMID: 35322372
- Tailoring Apixaban in Nanostructured Lipid Carrier Enhancing Its Oral Bioavailability and Anticoagulant Activity. | Zaky, MF., et al. 2022. Pharmaceutics. 15: PMID: 36678709
- Availability of Safe and Effective Therapeutic Options to Pregnant and Lactating Individuals Following the US FDA Pregnancy and Lactation Labeling Rule. | Patel, A., et al. 2023. J Pediatr.. PMID: 36806753
- A Sojourn on Liposomal Delivery System: Recent Advances and Future Prospects. | Kaur, S. and Singh, D. 2023. Assay Drug Dev Technol. 21: 48-64. PMID: 36856471
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Nintedanib esylate, 25 mg | sc-396761 | 25 mg | $87.00 | |||
Nintedanib esylate, 50 mg | sc-396761A | 50 mg | $114.00 | |||
Nintedanib esylate, 100 mg | sc-396761B | 100 mg | $146.00 | |||
Nintedanib esylate, 500 mg | sc-396761C | 500 mg | $312.00 | |||
Nintedanib esylate, 1 g | sc-396761D | 1 g | $426.00 |
