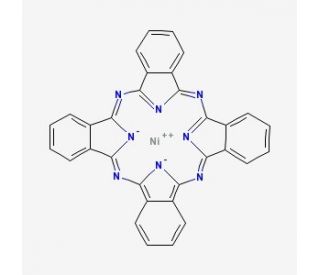
Nickel(II) phthalocyanine (CAS 14055-02-8)
QUICK LINKS
Nickel(II) phthalocyanine functions as a catalyst in various chemical reactions. Its mechanism of action involves facilitating the conversion of reactants into products by lowering the activation energy of the reaction. Nickel(II) phthalocyanine acts as a coordination complex, interacting with other molecules to form stable intermediates and transition states. Nickel(II) phthalocyanine participates in electron transfer processes, enabling the transfer of electrons between different species. Nickel(II) phthalocyanine also exhibits photochemical properties, allowing it to absorb light and undergo photochemical reactions. Nickel(II) phthalocyanine can act as a sensor for detecting specific analytes in solution, demonstrating its potential for use in analytical chemistry. Its unique molecular structure and electronic properties make it a versatile component in various experimental applications.
Nickel(II) phthalocyanine (CAS 14055-02-8) References
- Block copolymer micelles with near infrared metal phthalocyanine dyes for laser induced writing. | Acharya, H., et al. 2010. Macromol Rapid Commun. 31: 1071-7. PMID: 21590857
- Ultrafast charge transfer in nickel phthalocyanine probed by femtosecond Raman-induced Kerr effect spectroscopy. | Balakrishnan, G., et al. 2014. J Am Chem Soc. 136: 8746-54. PMID: 24841906
- Neutral nickel(II) phthalocyanine as a stable catalyst for visible-light-driven hydrogen evolution from water. | Yuan, YJ., et al. 2016. Dalton Trans. 45: 1359-63. PMID: 26743686
- A Simple Spectrofluorimetric Method Based on Quenching of a Nickel(II)-Phthalocyanine Complex to Determine Iron (III). | Çağlar, Y., et al. 2016. J Fluoresc. 26: 1381-9. PMID: 27256284
- Intermolecular interaction of nickel (ii) phthalocyanine tetrasulfonic acid tetrasodium salt with bovine serum albumin: A multi-technique study. | Dezhampanah, H., et al. 2017. Nucleosides Nucleotides Nucleic Acids. 36: 122-138. PMID: 27831822
- Synthesis, Spectral and Photophysical Properties of Anthracene Substituted Phthalocyanines; A Study as Polyurethane Electrospun Nanofibers. | Ku, KS., et al. 2018. J Nanosci Nanotechnol. 18: 1716-1722. PMID: 29448650
- A nickel(ii) complex under water-oxidation reaction: what is the true catalyst? | Feizi, H., et al. 2019. Dalton Trans. 48: 547-557. PMID: 30525137
- A new decomposition mechanism for metal complexes under water-oxidation conditions. | Najafpour, MM. and Feizi, H. 2019. Sci Rep. 9: 7483. PMID: 31097740
- Synthesis of an Activatable Tetra-Substituted Nickel Phthalocyanines-4(3H)-quinazolinone Conjugate and Its Antibacterial Activity. | Elsharif, AM., et al. 2019. Adv Pharmacol Sci. 2019: 5964687. PMID: 31143208
- Structure and electrochemical properties of carbon nanostructures derived from nickel(II) and iron(II) phthalocyanines. | Sanchez-Sanchez, A., et al. 2020. J Adv Res. 22: 85-97. PMID: 31956445
- How does mass transfer influence electrochemical carbon dioxide reduction reaction? A case study of Ni molecular catalyst supported on carbon. | Guo, C., et al. 2021. Chem Commun (Camb). 57: 1384-1387. PMID: 33438691
- Nickel phthalocyanine@graphene oxide/TiO2 as an efficient degradation catalyst of formic acid toward hydrogen production. | Keshipour, S. and Mohammad-Alizadeh, S. 2021. Sci Rep. 11: 16148. PMID: 34373517
- Harnessing Nickel Phthalocyanine-Based Electrochemical CNT Sponges for Ammonia Synthesis from Nitrate in Contaminated Water. | Li, Y., et al. 2022. ACS Appl Mater Interfaces. 14: 53884-53892. PMID: 36420862
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Nickel(II) phthalocyanine, 5 g | sc-269963 | 5 g | $90.00 |
