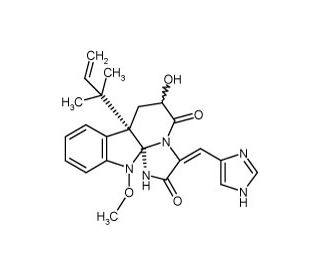
Neoxaline (CAS 71812-10-7)
See product citations (1)
QUICK LINKS
Neoxaline, as an indolotryptoline alkaloid, garners attention in the field of molecular biology for its role in natural product biosynthesis pathways. Researchers focus on its complex molecular scaffold to understand the genetic regulation and enzymatic steps involved in the formation of indole alkaloid structures within producing organisms. Specifically, neoxaline is a model compound in enzymology studies aiming to dissect the steps of tryptophan-derived alkaloid biosynthesis. In synthetic chemistry, it represents a challenging target for the total synthesis of indolotryptoline compounds, pushing the development of innovative synthetic routes that can manage the construction of its dense polycyclic system. These efforts not only advance the field of synthetic organic chemistry but also contribute to the discovery of methods that could be applied to the synthesis of related bioactive alkaloids. Additionally, in the study of plant secondary metabolites, neoxaline is used to explore plant defense mechanisms, providing insight into the ecological strategy of deterrence through chemical means.
Neoxaline (CAS 71812-10-7) References
- Combined molecular and biochemical approach identifies Aspergillus japonicus and Aspergillus aculeatus as two species. | Parenicová, L., et al. 2001. Appl Environ Microbiol. 67: 521-7. PMID: 11157212
- New Penicillium species associated with bulbs and root vegetables. | Overy, DP. and Frisvad, JC. 2003. Syst Appl Microbiol. 26: 631-9. PMID: 14666992
- Oxaline, a fungal alkaloid, arrests the cell cycle in M phase by inhibition of tubulin polymerization. | Koizumi, Y., et al. 2004. Biochim Biophys Acta. 1693: 47-55. PMID: 15276324
- A concise stereoselective route to the indoline spiroaminal framework of neoxaline and oxaline. | Sunazuka, T., et al. 2005. Org Lett. 7: 941-3. PMID: 15727480
- Efficient total synthesis of novel bioactive microbial metabolites. | Sunazuka, T., et al. 2008. Acc Chem Res. 41: 302-14. PMID: 18217720
- Two novel species of Aspergillus section Nigri from Thai coffee beans. | Noonim, P., et al. 2008. Int J Syst Evol Microbiol. 58: 1727-34. PMID: 18599725
- A branched biosynthetic pathway is involved in production of roquefortine and related compounds in Penicillium chrysogenum. | Ali, H., et al. 2013. PLoS One. 8: e65328. PMID: 23776469
- Asymmetric total synthesis of neoxaline. | Ideguchi, T., et al. 2013. J Am Chem Soc. 135: 12568-71. PMID: 23957424
- Novel key metabolites reveal further branching of the roquefortine/meleagrin biosynthetic pathway. | Ries, MI., et al. 2013. J Biol Chem. 288: 37289-95. PMID: 24225953
- Asymmetric Total Synthesis of Indole Alkaloids Containing an Indoline Spiroaminal Framework. | Yamada, T., et al. 2015. Chemistry. 21: 11855-64. PMID: 26147398
- Evolutionary formation of gene clusters by reorganization: the meleagrin/roquefortine paradigm in different fungi. | Martín, JF. and Liras, P. 2016. Appl Microbiol Biotechnol. 100: 1579-1587. PMID: 26668029
- Aspergillus labruscus sp. nov., a new species of Aspergillus section Nigri discovered in Brazil. | Fungaro, MHP., et al. 2017. Sci Rep. 7: 6203. PMID: 28740180
- Rapid Generation of Molecular Complexity by Chemical Synthesis: Highly Efficient Total Synthesis of Hexacyclic Alkaloid (-)-Chaetominine and Its Biosynthetic Implications. | Geng, H. and Huang, PQ. 2019. Chem Rec. 19: 523-533. PMID: 30252197
- Fungal metabolites: A recent trend and its potential biotechnological applications | Komal Agrawal, Pradeep Verma. 2021. New and Future Developments in Microbial Biotechnology and Bioengineering. 1-14.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Neoxaline, 1 mg | sc-281103 | 1 mg | $357.00 | |||
Neoxaline, 5 mg | sc-281103A | 5 mg | $561.00 |
