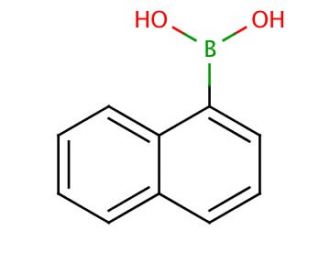
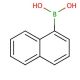
Molecular structure of Naphthalene-1-boronic acid, CAS Number: 13922-41-3
Naphthalene-1-boronic acid (CAS 13922-41-3)
Application:
Naphthalene-1-boronic acid is a naphthalene boronic acid building block
CAS Number:
13922-41-3
Purity:
≥98%
Molecular Weight:
171.99
Molecular Formula:
C10H9BO2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Naphthalene-1-boronic acid is a very useful building block for introduction of a naphthyl group through cross-coupling protocols. Naphthalene-1-boronic acid has been particularly useful in the synthesis of polyaromatic hydrocarbons utilizing the Suzuki reaction. This preparation of Naphthalene-1-boronic acid contains varying amounts of the cyclic anhydride form.
Naphthalene-1-boronic acid (CAS 13922-41-3) References
- A New and Concise Synthesis of 3-Hydroxybenzo[c]phenanthrene and 12-Hydroxybenzo[g]chrysene, Useful Intermediates for the Synthesis of Fjord-Region Diol Epoxides of Benzo[c]phenanthrene and Benzo[g]chrysene. | Kumar, S. 1997. J Org Chem. 62: 8535-8539. PMID: 11671997
- Efficient syntheses of C(8)-aryl adducts of adenine and guanine formed by reaction of radical cation metabolites of carcinogenic polycyclic aromatic hydrocarbons with DNA. | Dai, Q., et al. 2007. J Org Chem. 72: 4856-63. PMID: 17530898
- Novel [4 + 2]-benzannulation to access substituted benzenes and polycyclic aromatic and benzene-fused heteroaromatic compounds. | Raji Reddy, C., et al. 2014. Org Lett. 16: 3792-5. PMID: 25008279
- Versatile Self-Adapting Boronic Acids for H-Bond Recognition: From Discrete to Polymeric Supramolecules. | Georgiou, I., et al. 2017. J Am Chem Soc. 139: 2710-2727. PMID: 28051311
- Syntheses of adducts of active metabolites of carcinogenic polycyclic aromatic hydrocarbons with 2′-deoxyribonucleosides | Harvey, R. G., Dai, Q., Ran, C., Lim, K., Blair, I., & Penning, T. M. 2005. Polycyclic Aromatic Compounds. 25(5): 371-391.
- Blue organic light-emitting diode with improved color purity using 5-naphthyl-spiro[fluorene-7,9′-benzofluorene] | Jeon, S. O., Jeon, Y. M., Kim, J. W., Lee, C. W., & Gong, M. S. 2008. Organic electronics. 9(4): 522-532.
- Evaluation of Aromatic Boronic Acids as Ligands for Measuring Diabetes Markers on Carbon Nanotube Field‐Effect Transistors | Stefansson, S., Stefansson, L. A., Chung, S. W., Ko, K., Kwon, H. H., & Ahn, S. N. 2012. Journal of Nanotechnology. 2012(1): 371487.
- Bifunctional porous polymers bearing boronic and sulfonic acids for hydrolysis of cellulose | Yang, Q., & Pan, X. 2016. ACS Sustainable Chemistry & Engineering. 4(9): 4824-4830.
- A simple and highly selective fluorescent sensor for palladium based on benzofuran-2-boronic acid | Higashi, A., Kishikawa, N., Ohyama, K., & Kuroda, N. 2017. Tetrahedron Letters. 58(28): 2774-2778.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Naphthalene-1-boronic acid, 5 g | sc-255388 | 5 g | $79.00 |
