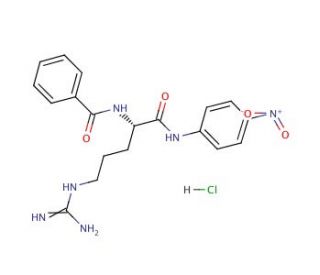
Nα-Benzoyl-L-arginine 4-nitroanilide hydrochloride (CAS 21653-40-7)
QUICK LINKS
Nα-Benzoyl-L-arginine 4-nitroanilide hydrochloride is a compound that functions as a substrate for the enzyme trypsin. When experimental applications, involved in the investigation of enzyme kinetics and the study of proteolytic activity. Its mode of action involves serving as a specific substrate for trypsin, which cleaves the compound at the arginine residue. This cleavage results in the release of 4-nitroaniline, allowing for the measurement of trypsin activity through spectrophotometric methods. By providing a specific substrate for trypsin, enables to study the enzyme′s catalytic efficiency and specificity in cleaving peptide bonds. Nα-Benzoyl-L-arginine 4-nitroanilide hydrochloride plays a functional role in elucidating the mechanisms of trypsin activity and its interaction with specific peptide sequences.
Nα-Benzoyl-L-arginine 4-nitroanilide hydrochloride (CAS 21653-40-7) References
- Feeding colostrum, its composition and feeding duration variably modify proliferation and morphology of the intestine and digestive enzyme activities of neonatal calves. | Blättler, U., et al. 2001. J Nutr. 131: 1256-63. PMID: 11285335
- Increased activity and stability of poly(ethylene glycol)-modified trypsin. | Gaertner, HF. and Puigserver, AJ. 1992. Enzyme Microb Technol. 14: 150-5. PMID: 1368397
- Identification of serine proteases from Leishmania braziliensis. | Guedes, HL., et al. 2007. Z Naturforsch C J Biosci. 62: 373-81. PMID: 17708443
- A chemical approach for detecting sulfenic acid-modified proteins in living cells. | Reddie, KG., et al. 2008. Mol Biosyst. 4: 521-31. PMID: 18493649
- Isolation, expression and characterization of a novel dual serine protease inhibitor, OH-TCI, from king cobra venom. | He, YY., et al. 2008. Peptides. 29: 1692-9. PMID: 18582511
- Efficient expression, purification and characterization of native human cystatin C in Escherichia coli periplasm. | Zhou, Y., et al. 2015. Protein Expr Purif. 111: 18-22. PMID: 25795130
- A flow-through nanoporous alumina trypsin bioreactor for mass spectrometry peptide fingerprinting. | Kjellander, M., et al. 2018. J Proteomics. 172: 165-172. PMID: 28942014
- Encapsulation and immobilization of ficin extract in electrospun polymeric nanofibers. | Rojas-Mercado, AS., et al. 2018. Int J Biol Macromol. 118: 2287-2295. PMID: 30031077
- Edema Induced by a Crotalus durissus terrificus Venom Serine Protease (Cdtsp 2) Involves the PAR Pathway and PKC and PLC Activation. | Costa, CRC., et al. 2018. Int J Mol Sci. 19: PMID: 30111691
- Vanillic acid as phospholipase A2 and proteases inhibitor: In vitro and computational analyses. | S Cesar, PH., et al. 2021. Biotechnol Appl Biochem. 68: 486-496. PMID: 32420666
- Expression of recombinant protease MarP from Mycobacterium tuberculosis in Pichia pastoris and its effect on human monocytes. | García-González, G., et al. 2021. Biotechnol Lett. 43: 1787-1798. PMID: 34028659
- Enzymatic Assays and Enzyme Histochemistry ofTuta absoluta Feeding on Tomato Leaves. | Hamza, R., et al. 2018. Bio Protoc. 8: e2993. PMID: 34395792
- The action of trypsin on synthetic chromogenic arginine substrates. | Somorin, O., et al. 1979. J Biochem. 85: 157-62. PMID: 762040
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Nα-Benzoyl-L-arginine 4-nitroanilide hydrochloride, 25 mg | sc-253173 | 25 mg | $84.00 | |||
Nα-Benzoyl-L-arginine 4-nitroanilide hydrochloride, 100 mg | sc-253173A | 100 mg | $235.00 |
