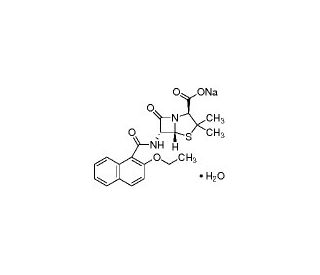
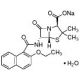
Nafcillin sodium salt monohydrate (CAS 7177-50-6)
QUICK LINKS
Nafcillin sodium salt monohydrate is a compound that is employed in research applications, especially in fields focusing on bacterial cell wall synthesis and antibiotic resistance mechanisms. Due to its stability against beta-lactamase enzymes produced by certain bacteria, it is a useful tool for studying the dynamics of antibiotic action and resistance in non-clinical models. Researchers utilize this sodium salt monohydrate form because of its increased solubility in aqueous solutions, which facilitates various laboratory experiments. Investigations using nafcillin sodium salt monohydrate focus on understanding how the beta-lactam ring of the molecule interacts with penicillin-binding proteins, which are critical for peptidoglycan synthesis in bacterial cell walls. Additionally, this compound aids in the exploration of the genetic and molecular factors that contribute to the emergence of resistance in bacterial populations.
Nafcillin sodium salt monohydrate (CAS 7177-50-6) References
- Synthesis and screening of a molecularly imprinted polymer library targeted for penicillin G. | Cederfur, J., et al. 2003. J Comb Chem. 5: 67-72. PMID: 12523836
- Development of a novel and automated fluorescent immunoassay for the analysis of beta-lactam antibiotics. | Benito-Peña, E., et al. 2005. J Agric Food Chem. 53: 6635-42. PMID: 16104778
- Determination of antimicrobials in sludge from infiltration basins at two artificial recharge plants by pressurized liquid extraction-liquid chromatography-tandem mass spectrometry. | Díaz-Cruz, MS., et al. 2006. J Chromatogr A. 1130: 72-82. PMID: 16822516
- QSRR analysis of β-lactam antibiotics on a penicillin G targeted MIP stationary phase. | Kempe, H. and Kempe, M. 2010. Anal Bioanal Chem. 398: 3087-96. PMID: 20936264
- Effect of various concentrations of antibiotics on osteogenic cell viability and activity. | Rathbone, CR., et al. 2011. J Orthop Res. 29: 1070-4. PMID: 21567453
- Synthesis of novel hapten and production of generic monoclonal antibody for immunoassay of penicillins residues in milk. | Jiao, SN., et al. 2013. J Environ Sci Health B. 48: 486-94. PMID: 23452214
- A controlled release of antibiotics from calcium phosphate-coated poly(lactic-co-glycolic acid) particles and their in vitro efficacy against Staphylococcus aureus biofilm. | Bastari, K., et al. 2014. J Mater Sci Mater Med. 25: 747-57. PMID: 24370968
- Rapid and easy multiresidue method for the analysis of antibiotics in meats by ultrahigh-performance liquid chromatography-tandem mass spectrometry. | Yamaguchi, T., et al. 2015. J Agric Food Chem. 63: 5133-40. PMID: 25656008
- A gold immunochromatographic assay for the rapid and simultaneous detection of fifteen β-lactams. | Chen, Y., et al. 2015. Nanoscale. 7: 16381-8. PMID: 26394361
- Removal and degradation of β-lactam antibiotics in water using didodecyldimethylammonium bromide-modified montmorillonite organoclay. | Saitoh, T. and Shibayama, T. 2016. J Hazard Mater. 317: 677-685. PMID: 27339949
- Production of anti-amoxicillin ScFv antibody and simulation studying its molecular recognition mechanism for penicillins. | Liu, J., et al. 2016. J Environ Sci Health B. 51: 742-50. PMID: 27383141
- Virtual mutation and directional evolution of anti-amoxicillin ScFv antibody for immunoassay of penicillins in milk. | He, X., et al. 2017. Anal Biochem. 517: 9-17. PMID: 27780696
- Development and validation of a quantitative confirmatory method for 30 β-lactam antibiotics in bovine muscle using liquid chromatography coupled to tandem mass spectrometry. | Di Rocco, M., et al. 2017. J Chromatogr A. 1500: 121-135. PMID: 28449875
- Improving the chromatographic selectivity of β-lactam residue analysis in milk using phenyl-column chemistry prior to detection by tandem mass spectrometry. | Di Rocco, M., et al. 2020. Anal Bioanal Chem. 412: 4461-4475. PMID: 32447520
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Nafcillin sodium salt monohydrate, 5 g | sc-236153 | 5 g | $140.00 |
