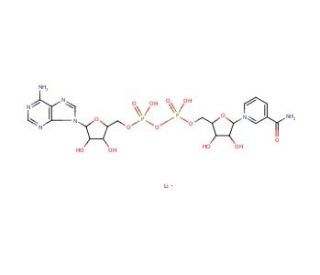
NAD+, Lithium Salt (CAS 64417-72-7)
QUICK LINKS
NAD+, Lithium Salt is a variant of nicotinamide adenine dinucleotide where lithium ions are used to neutralize the negative charge of the NAD+ molecule. In research, this salt form is particularly useful in enzymatic reactions and studies involving dehydrogenases, where it acts as a coenzyme essential for redox reactions. NAD+, Lithium Salt is used to investigate metabolic pathways and energy production, as it participates in the electron transport chain within cellular mitochondria. It is also instrumental in studies of DNA repair, given its role as a substrate for ADP-ribosyltransferases. Additionally, researchers utilize NAD+, Lithium Salt in assays to measure the activity of sirtuins and PARPs, which are important for understanding cellular regulation, stress responses, and the maintenance of genomic stability.
NAD+, Lithium Salt (CAS 64417-72-7) References
- Enzyme regulation by reversible zinc inhibition: glycerol phosphate dehydrogenase as an example. | Maret, W., et al. 2001. Chem Biol Interact. 130-132: 891-901. PMID: 11306104
- IFCC primary reference procedures for the measurement of catalytic activity concentrations of enzymes at 37 degrees C. Part 3. Reference procedure for the measurement of catalytic concentration of lactate dehydrogenase. | Schumann, G., et al. 2002. Clin Chem Lab Med. 40: 643-8. PMID: 12211663
- l- and d-Lactate assay in real milk samples with immobilized enzyme reactors and graphite electrode. | Marrazza, G., et al. 1994. Talanta. 41: 1007-14. PMID: 18966029
- IFCC reference procedures for measurement of the catalytic concentrations of enzymes: corrigendum, notes and useful advice. International Federation of Clinical Chemistry and Laboratory Medicine (IFCC)--IFCC Scientific Division. | Schumann, G., et al. 2010. Clin Chem Lab Med. 48: 615-21. PMID: 20298135
- Probing lactate dehydrogenase activity in tumors by measuring hydrogen/deuterium exchange in hyperpolarized l-[1-(13)C,U-(2)H]lactate. | Kennedy, BW., et al. 2012. J Am Chem Soc. 134: 4969-77. PMID: 22316419
- Reversal of part of the aldehyde dehydrogenase reaction pathway during the hydrolysis of an ester. | Duncan, RJ. 1979. Biochem J. 183: 459-62. PMID: 230823
- High- and low-affinity PEGylated hemoglobin-based oxygen carriers: Differential oxidative stress in a Guinea pig transfusion model. | Alomari, E., et al. 2018. Free Radic Biol Med. 124: 299-310. PMID: 29920341
- Structure of adenosine-5'-mononicotinate (AMN) trihydrate: an analog of NAD for testing intramolecular stacking. | Ramasubbu, N. and Parthasarathy, R. 1988. Acta Crystallogr C. 44 (Pt 12): 2140-4. PMID: 3270554
- Simvastatin Impairs Glucose Homeostasis in Mice Depending on PGC-1α Skeletal Muscle Expression. | Panajatovic, MV., et al. 2020. Biomedicines. 8: PMID: 32942550
- Modification of mouse testicular lactate dehydrogenase by pyridoxal 5'-phosphate. | Gould, KG. and Engel, PC. 1980. Biochem J. 191: 365-71. PMID: 6786279
- Pyruvate decarboxylase catalyzes decarboxylation of branched-chain 2-oxo acids but is not essential for fusel alcohol production by Saccharomyces cerevisiae. | ter Schure, EG., et al. 1998. Appl Environ Microbiol. 64: 1303-7. PMID: 9546164
- An enzymatic method for oxalate automated using the Hitachi 911 analyzer. | Langman, LJ. and Allen, LC. 1998. Clin Biochem. 31: 429-32. PMID: 9721445
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
NAD+, Lithium Salt, 1 g | sc-208085 | 1 g | $272.00 |
