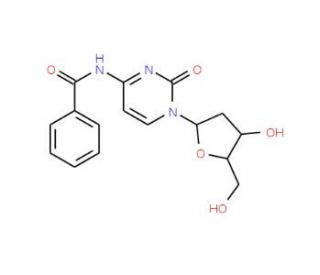
Molecular structure of N4-Benzoyl-2′-deoxycytidine, CAS Number: 4836-13-9
N4-Benzoyl-2′-deoxycytidine (CAS 4836-13-9)
Alternate Names:
N-Benzoyl-2-Deoxy-Cytidine; NBz-2′-dC
CAS Number:
4836-13-9
Molecular Weight:
331.32
Molecular Formula:
C16H17N3O5
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
N4-Benzoyl-2′-deoxycytidine, a synthetic nucleoside analog derived from natural deoxycytidine, holds significance as a tool in biochemistry and molecular biology research. It has been employed in studying DNA and RNA, including their structure, function, and regulatory mechanisms. Furthermore, N4-Benzoyl-2′-deoxycytidine has been instrumental in investigating gene expression, deciphering protein-DNA interactions, and unraveling the intricate mechanisms governing DNA replication and repair.
N4-Benzoyl-2′-deoxycytidine (CAS 4836-13-9) References
- Rapid and selective reduction of amide group by borane-amine complexes in acyl protected nucleosides. | Sergueeva, ZA., et al. 2000. Nucleosides Nucleotides Nucleic Acids. 19: 275-82. PMID: 10772715
- Formation of N-branched oligonucleotides as by-products in solid-phase oligonucleotide synthesis. | Cazenave, C., et al. 2006. Oligonucleotides. 16: 181-5. PMID: 16764541
- Convenient syntheses of 3'-amino-2',3'-dideoxynucleosides, their 5'-monophosphates, and 3'-aminoterminal oligodeoxynucleotide primers. | Eisenhuth, R. and Richert, C. 2009. J Org Chem. 74: 26-37. PMID: 19053612
- Studies on 2',3'-dideoxy-2',3'-didehydropyrimidine nucleosides. II. N4-benzoyl-2',3'-dideoxy-2',3'-didehydrocytidine as a prodrug of 2',3'-dideoxy-2',3'-didehydrocytidine (DDCN). | Kawaguchi, T., et al. 1989. Chem Pharm Bull (Tokyo). 37: 2547-9. PMID: 2557984
- Synthesis and selective cleavage of oligodeoxyribonucleotides containing non-chiral internucleotide phosphoramidate linkages. | Mag, M. and Engels, JW. 1989. Nucleic Acids Res. 17: 5973-88. PMID: 2771637
- Synthetic nucleosides and nucleotides. XXI. On the synthesis and biological evaluations of 2'-deoxy-alpha-D-ribofuranosyl nucleosides and nucleotides. | Yamaguchi, T. and Saneyoshi, M. 1984. Chem Pharm Bull (Tokyo). 32: 1441-50. PMID: 6467456
- Synthesis and binding properties of oligodeoxynucleotides containing phenylphosphon(othio)ate linkages. | Mag, M., et al. 1997. Bioorg Med Chem. 5: 2213-20. PMID: 9459019
- Nucleotides. Part XXVII† Bis[2-(p-nitrophenyl)ethyl] Phosphorochloridate, a New Versatile Phosphorylating Agent in Nucleotide Chemistry | Frank Himmelsbach, Ramamurthy Charubala, Wolfgang Pfleiderer. 1987. Helvetica Chimica Acta. 70: 1286-1295.
- Engineering tethered DNA molecules by the convertible nucleoside approach | Andrew M Macmillan, Gregory L Verdine ∗. 1991. Tetrahedron. 47: 2603-2616.
- Novel approach to diastereoselective synthesis of 2′-deoxy[5′-2H1]ribonucleoside derivatives by reduction of the corresponding 5′-O-acetyl-2′-deoxy-5′-phenylselenoribonucleoside derivatives with a Bu3Sn2H-Et3B system | Etsuko Kawashima, Keizo Toyama, Kenshiro Ohshima, Masatsune Kainosho, Yoshimasa Kyogoku, Yoshiharu Ishido. 1997. Chirality. 9: 435-442.
- Solid phase glycosidation of oligonucleotides | Matteo Adinolfi, Gaspare Barone, Lorenzo De Napoli, Luigi Guariniello, Alfonso Iadonisi ∗, Gennaro Piccialli. 1999. Tetrahedron Letters. 40: 2607-2610.
- New Types of Very Efficient Photolabile Protecting Groups Based upon the [2-(2-Nitrophenyl)propoxy]carbonyl (NPPOC) Moiety | Sigrid Bühler, Irene Lagoja, Heiner Giegrich, Klaus-Peter Stengele, Wolfgang Pfleiderer. 2004. Helvetica Chimica Acta. 87: 620-659.
- Electron-deficient benzotriazoles for the selective N-acetylation of nucleosides | Andrew K. Reid, Callum J. McHugh, Graham Richie, Duncan Graham. 2006. Tetrahedron Letters. 47: 4201-4203.
- A Simple and Convenient Method for the Selective N-Acylations of Cytosine Nucleosides | V. Bhat, B. G. Ugarkar, V. A. Sayeed, K. Grimm, N. Kosora, P. A. Domenico & show all. 2006. Nucleosides and Nucleotides. 8: 179-183.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N4-Benzoyl-2′-deoxycytidine, 5 g | sc-295827 | 5 g | $122.00 | |||
N4-Benzoyl-2′-deoxycytidine, 25 g | sc-295827A | 25 g | $316.00 |
