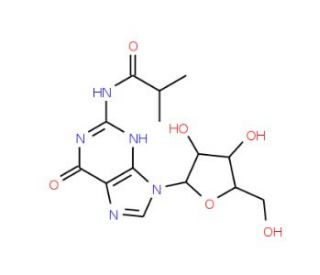
N2-Isobutyrylguanosine (CAS 64350-24-9)
QUICK LINKS
N2-Isobutyrylguanosine is a chemical compound that functions as a modulator of gene expression. It acts by participating in the process of RNA modification, in the methylation of RNA molecules. N2-Isobutyrylguanosine plays a role in regulating the stability and translation of mRNA, thereby influencing protein synthesis within the cell. N2-Isobutyrylguanosine achieves this by interacting with specific enzymes involved in RNA modification, ultimately impacting the gene expression profile. Its mode of action involves the modification of guanosine residues within RNA, leading to alterations in the structure and function of the RNA molecule. N2-Isobutyrylguanosine′s function in gene expression regulation is of interest in various development endeavors, as it provides insight into the molecular mechanisms underlying cellular processes.
N2-Isobutyrylguanosine (CAS 64350-24-9) References
- Chemical syntheses of inhibitory substrates of the RNA-RNA ligation reaction catalyzed by the hairpin ribozyme. | Massey, AP. and Sigurdsson, ST. 2004. Nucleic Acids Res. 32: 2017-22. PMID: 15064361
- New synthetic routes to protected purine 2'-O-methylriboside-3'-O-phosphoramidites using a novel alkylation procedure. | Sproat, BS., et al. 1990. Nucleic Acids Res. 18: 41-9. PMID: 2308835
- Phosphorothioate analogs of P1,P3-di(nucleosid-5'-yl) triphosphates: Synthesis, assignment of the absolute configuration at P-atoms and P-stereodependent recognition by Fhit hydrolase. | Kaczmarek, R., et al. 2016. Bioorg Med Chem. 24: 5068-5075. PMID: 27591011
- Catalytic asymmetric and stereodivergent oligonucleotide synthesis. | Featherston, AL., et al. 2021. Science. 371: 702-707. PMID: 33574208
- Quick Access to Nucleobase-Modified Phosphoramidites for the Synthesis of Oligoribonucleotides Containing Post-Transcriptional Modifications and Epitranscriptomic Marks. | Ziemkiewicz, K., et al. 2022. J Org Chem. 87: 10333-10348. PMID: 35857285
- Synthesis of purine derivatives of Me-TaNA and properties of Me-TaNA-modified oligonucleotides. | Fuchi, Y., et al. 2023. Org Biomol Chem. 21: 5203-5213. PMID: 37309204
- Comparative cytostatic activity of different antiherpetic drugs against herpes simplex virus thymidine kinase gene-transfected tumor cells. | Balzarini, J., et al. 1994. Mol Pharmacol. 45: 1253-8. PMID: 8022417
- Selective 5′-O-Acetylation of 2′-Deoxynucleosides and Nucleosides by a Modified Mitsunobu Procedure | Gong-Xin He &Norbert Bischofberger. 1997 -. Nucleosides and Nucleotides. Volume 16, Issue 3: Pages 257-263.
- An Efficient Multigram Synthesis of Monomers for the Preparation of Novel Oligonucleotides Containing Isosteric Non-Phosphorous Backbones | Stuart Dimock,Balkrishen Bhat,Didier Peoc'h,Yogesh S. Sanghvi &Eric E. Swayze. 1997. Nucleosides and Nucleotides. Volume 16, - Issue 7-9: Pages 1629-1632.
- Nucleic Acid Triple Helices: Stability Effects of Nucleobase Modifications | Robles, J.; Grandas, A.; Pedroso, E.; Luque, F.; Eritja, R.; Orozco, M. 2002,. Current Organic Chemistry,. Volume 6, Number 14,: pp. 1333-1368(36).
- Rapid identification of short oligonucleotide impurities using lithium adduct consolidated MALDI-TOF mass spectrometry | OB Becette, A Tran, JP Marino, JW Jones… - International Journal of …, 2022 - Elsevier. November 2022,. International Journal of Mass Spectrometry. Volume 481,: 116913.
- Synthesis and hybridization study of a boranophosphate-linked oligothymidine deoxynucleotide | J Zhang, T Terhorst, MD Matteucci - Tetrahedron letters, 1997 - Elsevier. 14 July 1997,. Tetrahedron Letters. Volume 38, Issue 28,: Pages 4957-4960.
- A convenient preparation of protected 3′-deoxyguanosine from guanosine | GX He, N Bischofberger - Tetrahedron letters, 1995 - Elsevier. 25 September 1995,. Tetrahedron Letters. Volume 36, Issue 39,: Pages 6991-6994.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N2-Isobutyrylguanosine, 5 g | sc-286463 | 5 g | $70.00 | |||
N2-Isobutyrylguanosine, 10 g | sc-286463A | 10 g | $94.00 |
