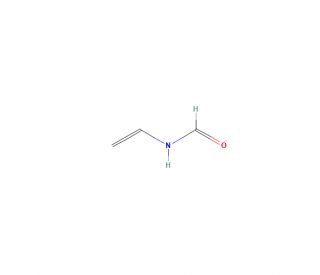
N-Vinylformamide (CAS 13162-05-5)
QUICK LINKS
N-Vinylformamide, a liquid varying from colorless to yellow, is soluble in several organic solvents such as water, ethanol, and chloroform. It is recognized for its high reactivity, and has various applications across chemistry, biology, and materials science. The molecule has a polar, unsaturated structure that makes it an excellent reactive monomer for various polymerization reactions. N-Vinylformamide is identified as a promising reactive monomer in the development of biomaterials like hydrogels and microcapsules and has been explored as a co-monomer alongside other vinyl monomers. Its applications extend to various scientific explorations, encompassing the fabrication of polymers, adhesives, coatings, and additional materials. It is also used as a reactive monomer in organic synthesis and as a crosslinker for hydrogels.
N-Vinylformamide (CAS 13162-05-5) References
- Enzyme-containing Michael-adduct-based coatings. | Drevon, GF., et al. 2003. Biomacromolecules. 4: 675-82. PMID: 12741784
- Poly(N-vinylformamide)-A drag-reducing polymer for biomedical applications. | Marhefka, JN., et al. 2006. Biomacromolecules. 7: 1597-603. PMID: 16677044
- Synthesis and characterization of a novel graft copolymer of partially carboxymethylated guar gum and N-vinylformamide. | Mishra, MM., et al. 2015. Carbohydr Polym. 115: 776-84. PMID: 25439961
- Study of cellulolytic enzyme immobilization on copolymers of N-vinylformamide. | Tąta, A., et al. 2015. Spectrochim Acta A Mol Biomol Spectrosc. 149: 494-504. PMID: 25978017
- Hydrogen-Bonded Multilayer Films Based on Poly(N-vinylamide) Derivatives and Tannic Acid. | Takemoto, Y., et al. 2015. Langmuir. 31: 6863-9. PMID: 26052735
- Aggregation Control by Multi-stimuli-Responsive Poly(N-vinylamide) Derivatives in Aqueous System. | Kawatani, R., et al. 2017. Nanoscale Res Lett. 12: 461. PMID: 28738665
- Thermal Treatment of Poly (N-vinylformamide) Produced Hydrogels Without the Use of Chemical Crosslinkers. | Ajiro, H., et al. 2017. J Nanosci Nanotechnol. 17: 837-41. PMID: 29634180
- Synthesis of Poly(N-vinylamide)s and Poly(vinylamine)s and Their Block Copolymers by Organotellurium-Mediated Radical Polymerization. | Fan, W. and Yamago, S. 2019. Angew Chem Int Ed Engl. 58: 7113-7116. PMID: 30920088
- Poly(vinylamine-co-N-isopropylacrylamide) linear polymer and hydrogels with tuned thermoresponsivity. | Fischer, T., et al. 2020. Soft Matter. 16: 6549-6562. PMID: 32617537
- Ultrahigh Flux and Strong Affinity Poly(N-vinylformamide)-Grafted Polypropylene Membranes for Continuous Removal of Organic Micropollutants from Water. | Gui, Q., et al. 2021. ACS Appl Mater Interfaces. 13: 20796-20809. PMID: 33884869
- Tailor-Made Poly(vinylamine) via Purple LED-Activated RAFT Polymerization of N-vinylformamide. | Kurowska, I., et al. 2023. Macromol Rapid Commun. 44: e2200729. PMID: 36443826
- Injectable Colloidal Hydrogels of N-Vinylformamide Microgels Dispersed in Covalently Interlinked pH-Responsive Methacrylic Acid-Based Microgels. | Wang, X., et al. 2023. Biomacromolecules. 24: 2173-2183. PMID: 37026759
- Homo- and Copolymer Hydrogels Based on N-Vinylformamide: An Investigation of the Impact of Water Structure on Controlled Release. | Yooyod, M., et al. 2023. Gels. 9: PMID: 37102945
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Vinylformamide, 25 g | sc-476794 | 25 g | $42.00 |
