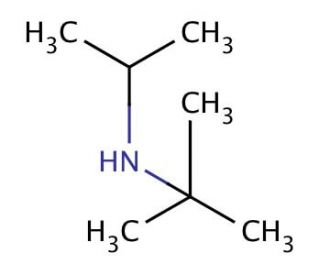
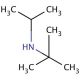
Molecular structure of N-tert-Butylisopropylamine, CAS Number: 7515-80-2
N-tert-Butylisopropylamine (CAS 7515-80-2)
CAS Number:
7515-80-2
Molecular Weight:
115.22
Molecular Formula:
C7H17N
Supplemental Information:
This is as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
N-tert-Butylisopropylamine functions as a catalyst in various chemical reactions. It acts as a base in organic chemistry, facilitating the formation of carbon-carbon and carbon-nitrogen bonds through its nucleophilic properties. N-tert-Butylisopropylamine′s mechanism of action involves its interaction with acidic substrates, leading to the formation of stable intermediates that drive the desired chemical transformations.
N-tert-Butylisopropylamine (CAS 7515-80-2) References
- Determination of conformer-specific partition coefficients in octanol/water systems. | Kraszni, M., et al. 2003. J Med Chem. 46: 2241-5. PMID: 12747795
- Synthesis and C-alkylation of hindered aldehyde enamines. | Hodgson, DM., et al. 2009. J Org Chem. 74: 1019-28. PMID: 19105636
- Aerobic oxidative N-dealkylation of tertiary amines in aqueous solution catalyzed by rhodium porphyrins. | Ling, Z., et al. 2013. Chem Commun (Camb). 49: 4214-6. PMID: 23223161
- Sterically hindered malonamide monomers for the step growth synthesis of polyesters and polyamides. | Tyler, SN. and Webster, RL. 2014. Chem Commun (Camb). 50: 10665-8. PMID: 25078455
- Semi-heterogeneous Dual Nickel/Photocatalysis using Carbon Nitrides: Esterification of Carboxylic Acids with Aryl Halides. | Pieber, B., et al. 2019. Angew Chem Int Ed Engl. 58: 9575-9580. PMID: 31050132
- Semiheterogeneous Dual Nickel/Photocatalytic (Thio)etherification Using Carbon Nitrides. | Cavedon, C., et al. 2019. Org Lett. 21: 5331-5334. PMID: 31247752
- Evidence for Photocatalyst Involvement in Oxidative Additions of Nickel-Catalyzed Carboxylate O-Arylations. | Malik, JA., et al. 2020. J Am Chem Soc. 142: 11042-11049. PMID: 32469219
- Nickel-Catalyzed C-Heteroatom Cross-Coupling Reactions under Mild Conditions via Facilitated Reductive Elimination. | Zhu, C., et al. 2021. Angew Chem Int Ed Engl. 60: 17810-17831. PMID: 33252192
- Heterogeneous metallaphotoredox catalysis in a continuous-flow packed-bed reactor. | Hsu, WH., et al. 2022. Beilstein J Org Chem. 18: 1123-1130. PMID: 36105732
- On-Demand Release of Secondary Amine Bases for the Activation of Catalysts and Crosslinkers. | Spitzbarth, B. and Eelkema, R. 2023. Chemistry. 29: e202203028. PMID: 36541271
- A novel atom-efficient, one-pot synthesis of sulfonylguanidines and sulfamoylguanidines | Jos H.M. Lange, Stefan Verhoog, Hans J. Sanders, Arnold van Loevezijn, Chris G. Kruse. 2011. https://doi.org/10.1016/j.tetlet.2011.04.031. 52: 3198-3200.
- Overcoming limitations in dual photoredox/nickel-catalysed C–N cross-couplings due to catalyst deactivation | Sebastian Gisbertz, Susanne Reischauer & Bartholomäus Pieber. 2020. Nature Catalysis. 3: pages 611–620.
- Modulating the Surface and Photophysical Properties of Carbon Dots to Access Colloidal Photocatalysts for Cross-Couplings | Zhouxiang Zhao, Bartholomäus Pieber*, and Martina Delbianco*, et al. 2022. ACS Catal. 12: 13831–13837.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-tert-Butylisopropylamine, 5 g | sc-257868 | 5 g | $105.00 |
