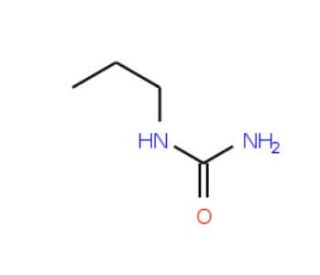
N-propylurea (CAS 627-06-5)
QUICK LINKS
N-Propylurea (NPU) is a urea derivative that holds significance in organic chemistry. This is a colorless, odorless, and crystalline solid. N-Propylurea serves as a versatile tool in scientific research, acting as a reagent, catalyst, and solvent. Its role as a reagent is in synthesizing diverse organic compounds, such as dyes, and agrochemicals. Additionally, N-Propylurea serves as a catalyst in facilitating numerous organic reactions, including the Knoevenagel condensation, Michael addition, and aldol condensation. Furthermore, N-Propylurea proves valuable as a solvent for various organic reactions, like amine acylation and ester hydrolysis. In organic reactions, N-propylurea functions as a nucleophile, engaging electrophilic centers within substrates. It also exhibits characteristics of a base catalyst, enabling the deprotonation of protic substrates and promoting the formation of new covalent bonds. Moreover, N-Propylurea′s ability to form hydrogen bonds with substrates contributes to the facilitation of substrate reactivity with other reagents.
N-propylurea (CAS 627-06-5) References
- Photoproducts in DNA irradiated in vitro and in vivo under extreme environmental conditions. | Riklis, E. 1989. Adv Space Res. 9: 223-32. PMID: 11537296
- Diuretic effects of some mercurated substituted N-n-propylurea derivatives. | ROSEN, H., et al. 1957. Proc Soc Exp Biol Med. 95: 635-6. PMID: 13465752
- Discovery of N-propylurea 3-benzylpiperidines as selective CC chemokine receptor-3 (CCR3) antagonists. | Varnes, JG., et al. 2004. Bioorg Med Chem Lett. 14: 1645-9. PMID: 15026042
- Synthesis and biological evaluation of new symmetrical derivatives as cytotoxic agents and apoptosis inducers. | Sanmartín, C., et al. 2005. Bioorg Med Chem. 13: 2031-44. PMID: 15727857
- Pseudomonas aeruginosa cytochrome c551 denaturation by five systematic urea derivatives that differ in the alkyl chain length. | Kobayashi, S., et al. 2017. Biosci Biotechnol Biochem. 81: 1274-1278. PMID: 28318436
- Energy/Electron Transfer Switch for Controlling Optical Properties of Silicon Quantum Dots. | Abdelhameed, M., et al. 2018. Sci Rep. 8: 17068. PMID: 30459354
- [Induction of tumours by different endogenously formed N-nitroso-ureas in the hooded rat (author's transl)]. | Schneider, J., et al. 1977. Zentralbl Allg Pathol. 121: 61-75. PMID: 322421
- The selective photolysis of dihydrothymidine. | Kondo, Y. and Witkop, B. 1968. J Am Chem Soc. 90: 3258-9. PMID: 5649186
- Selective assay procedure for chlorpropamide in the presence of its decomposition products. | Kaistha, KK. 1969. J Pharm Sci. 58: 235-7. PMID: 5779868
- Skin tumors induced by painting nitrosoalkylureas on mouse skin. | Lijinsky, W. and Winter, C. 1981. J Cancer Res Clin Oncol. 102: 13-20. PMID: 7334051
- Identification of alkylureas after nitrosation-denitrosation of a bonito fish product, crab, lobster, and bacon. | Mirvish, SS., et al. 1980. J Agric Food Chem. 28: 1175-82. PMID: 7451744
- Solution Properties of Urea and Its Derivatives in Water: Evidence from Ultrasonic Relaxation Spectra | A. Rupprecht and U. Kaatze. 2002. J. Phys. Chem. A., 106, 38,: 8850–8858.
- Effect of temperature and ionic strength on volumetric and acoustic properties of solutions of urea alkyl derivatives in aqueous NaCl | J Krakowiak, J Wawer - The Journal of Chemical Thermodynamics, 2015 - Elsevier. November 2015,. The Journal of Chemical Thermodynamics. Volume 90,: Pages 232-241.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-propylurea, 5 g | sc-269891 | 5 g | $62.00 |
