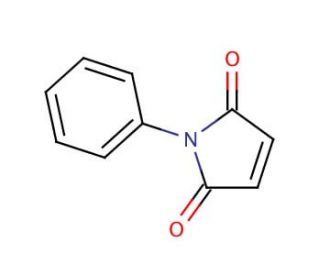
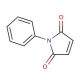
N-Phenylmaleimide (CAS 941-69-5)
QUICK LINKS
N-Phenylmaleimide is a chemical compound utilized as a reagent in the field of polymer chemistry and materials science. It is known for its ability to undergo Diels-Alder reactions with dienes, which is used to modify the properties of polymers or create new polymeric materials with enhanced characteristics. In research, N-Phenylmaleimide is employed to study the cross-linking of elastomers, where it improves the thermal stability and mechanical strength of the resulting materials. Additionally, it serves as a functional monomer in copolymerization reactions to introduce phenyl rings into polymer backbones, which can affect the electronic properties of the polymers. The reactivity of N-Phenylmaleimide with thiol groups also makes it valuable in the modification of proteins and peptides, which is important for understanding protein structure and function in a non-biological context.
N-Phenylmaleimide (CAS 941-69-5) References
- Mechanism of migration of the trimethylsilyl group during reactions of methoxy[(trimethylsilyl)ethoxy]carbene with N-phenylmaleimide and C(60). | Sharma, PK., et al. 2001. J Org Chem. 66: 7496-9. PMID: 11681966
- Vibrational spectroscopy of N-phenylmaleimide. | Parker, SF. 2006. Spectrochim Acta A Mol Biomol Spectrosc. 63: 544-9. PMID: 16157505
- Diastereo- and enantioselective [3 + 2] cycloaddition reaction of Morita-Baylis-Hillman carbonates of isatins with N-phenylmaleimide catalyzed by Me-DuPhos. | Wang, Y., et al. 2012. J Org Chem. 77: 4143-7. PMID: 22471756
- Synthesis and characterization of novel bioactive 1,2,4-oxadiazole natural product analogs bearing the N-phenylmaleimide and N-phenylsuccinimide moieties. | Maftei, CV., et al. 2013. Beilstein J Org Chem. 9: 2202-15. PMID: 24222789
- The effects of sample handling and N-phenylmaleimide on concentration of adrenocorticotrophic hormone in equine plasma. | Rendle, DI., et al. 2015. Equine Vet J. 47: 587-91. PMID: 24980684
- RAFT Dispersion Alternating Copolymerization of Styrene with N-Phenylmaleimide: Morphology Control and Application as an Aqueous Foam Stabilizer. | Yang, P., et al. 2016. Macromolecules. 49: 6731-6742. PMID: 27708458
- Interactions of sodium-proton exchange mechanism in dog red blood cells with N-phenylmaleimide. | Parker, JC. and Glosson, PS. 1987. Am J Physiol. 253: C60-5. PMID: 3037914
- Facile fabrication of durable antibacterial and anti-felting wool fabrics with enhanced comfort via novel N-phenylmaleimide finishing. | Liu, G., et al. 2022. Bioprocess Biosyst Eng. 45: 921-929. PMID: 35348877
- Antibacterial Mechanism of N-PMI and the Characteristics of PMMA-Co-N-PMI Copolymer. | Shi, H., et al. 2022. Chem Biodivers. 19: e202100753. PMID: 35560720
- Radical reaction extrusion copolymerization mechanism of MMA and N-phenylmaleimide and properties of products. | Shi, H., et al. 2022. RSC Adv. 12: 26251-26263. PMID: 36199612
- Exploring the peri- and stereo- selectivities of the cycloaddition reaction of 2-(2- dimethylaminovinyl)-1-benzopyran-4-one with N-phenylmaleimide (NPM) and dimethylacetylenedicarboxylate (DMAD) - A DFT study. | Arhin, R., et al. 2023. J Mol Graph Model. 121: 108451. PMID: 36924702
- The site and stoichiometry of the N-phenylmaleimide reaction with myosin when weakly-binding crossbridges are formed in skinned rabbit psoas fibers. | Ehrlich, A., et al. 1995. Biochim Biophys Acta. 1232: 13-20. PMID: 7495834
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Phenylmaleimide, 5 g | sc-250486A | 5 g | $27.00 | |||
N-Phenylmaleimide, 25 g | sc-250486 | 25 g | $56.00 | |||
N-Phenylmaleimide, 100 g | sc-250486B | 100 g | $114.00 | |||
N-Phenylmaleimide, 500 g | sc-250486C | 500 g | $252.00 |
