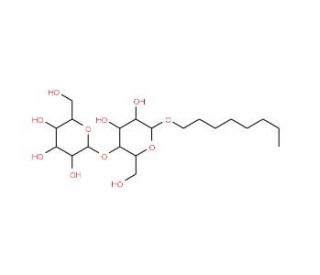
n-Octyl-β-D-maltopyranoside (CAS 82494-08-4)
QUICK LINKS
n-Octyl-β-D-maltopyranoside is a valuable chemical compound extensively utilized in biochemical and biophysical research due to its unique properties and versatile applications. Its mechanism of action involves acting as a non-ionic detergent, disrupting hydrophobic interactions, and facilitating the solubilization and stabilization of membrane proteins. This detergent has been widely employed in various experimental techniques, including protein extraction, purification, and crystallization. Researchers utilize n-Octyl-β-D-maltopyranoside to solubilize integral membrane proteins, enabling their study and characterization in vitro. Moreover, it has been instrumental in the preparation of lipid-protein complexes and reconstitution of membrane proteins into artificial lipid bilayers for biophysical studies. Its mild detergent properties make it particularly suitable for maintaining the native structure and function of membrane proteins, crucial for elucidating their biological roles and mechanisms of action. Ongoing research continues to explore novel applications of n-Octyl-β-D-maltopyranoside in structural biology, drug discovery, and understanding cellular processes at the molecular level.
n-Octyl-β-D-maltopyranoside (CAS 82494-08-4) References
- Crystallization and preliminary X-ray analysis of the trehalose/maltose ABC transporter MalFGK2 from Thermococcus litoralis. | Schiefner, A., et al. 2002. Acta Crystallogr D Biol Crystallogr. 58: 2147-9. PMID: 12454482
- Structures of micelles formed by synthetic alkyl glycosides with unsaturated alkyl chains. | Milkereit, G., et al. 2005. J Colloid Interface Sci. 284: 704-13. PMID: 15780314
- Haemolytic activity of maltopyranoside surfactants. | Söderlind, E. and Karlsson, L. 2006. Eur J Pharm Biopharm. 62: 254-9. PMID: 16274978
- Modeling the micellization behavior of mixed and pure n-alkyl-maltosides. | Tsamaloukas, AD., et al. 2009. Langmuir. 25: 4393-401. PMID: 19366219
- Nanocomposite formation between alpha-glucosyl stevia and surfactant improves the dissolution profile of poorly water-soluble drug. | Uchiyama, H., et al. 2012. Int J Pharm. 428: 183-6. PMID: 22265914
- Tuning micelle dimensions and properties with binary surfactant mixtures. | Oliver, RC., et al. 2014. Langmuir. 30: 13353-61. PMID: 25312254
- Successful amphiphiles as the key to crystallization of membrane proteins: Bridging theory and practice. | Breibeck, J. and Rompel, A. 2019. Biochim Biophys Acta Gen Subj. 1863: 437-455. PMID: 30419284
- Capillary electrophoresis of herbicides. III. Evaluation of octylmaltopyranoside chiral surfactant in the enantiomeric separation of phenoxy acid herbicides. | Mechref, Y. and el Rassi, Z. 1996. Chirality. 8: 518-524. PMID: 8970750
- Comparison of alkylglycoside surfactants in enantioseparation by capillary electrophoresis. | Mechref, Y. and El Rassi, Z. 1997. Electrophoresis. 18: 912-8. PMID: 9221877
- Complex effect of ethyl branching on the supramolecular structure of a long chain neoglycolipid | Milkereit, G., & Garamus, V. M. 2005. Colloids and Surfaces A: Physicochemical and Engineering Aspects. 268(1-3): 155-161.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
n-Octyl-β-D-maltopyranoside, 1 g | sc-281089 | 1 g | $676.00 |
