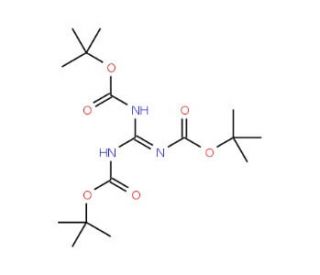
N,N′,N′′-Tri-Boc-guanidine (CAS 216584-22-4)
QUICK LINKS
N,N′,N′′-Tri-Boc-guanidine is a valuable tri-alkylated guanidine derivative extensively employed in organic synthesis. This white solid readily dissolves in organic solvents at room temperature, making it highly convenient to work with. Its versatility has led to widespread use as an intermediate in crafting an array of compounds, including agrochemicals and specialty chemicals. One of its primary applications lies in peptide and peptidomimetic synthesis, thanks to its exceptional stability and reactivity. Moreover, it plays a role in generating peptidomimetic libraries for drug discovery. Beyond that, N,N′,N′′-Tri-Boc-guanidine serves as a reagent in synthesizing various compounds, such as heterocycles, peptides, and peptidomimetics. Functioning as a nucleophile during organic synthesis, N,N′,N′′-Tri-Boc-guanidine eagerly interacts with electrophiles like carbonyl compounds and halides, resulting in the formation of amides and other derivatives. Additionally, it showcases its prowess by engaging carbon-carbon double bonds, leading to the creation of cyclic structures. In essence, N,N′,N′′-Tri-Boc-guanidine proves to be an invaluable asset in the realm of organic chemistry, facilitating the creation of diverse compounds with efficiency and precision. Its wide-ranging applications make it an essential component in the pursuit of scientific advancements.
N,N′,N′′-Tri-Boc-guanidine (CAS 216584-22-4) References
- Synthesis of 2'-O-modified adenosine building blocks and application for RNA interference. | Odadzic, D., et al. 2008. Bioorg Med Chem. 16: 518-29. PMID: 17904849
- Peptide ligation-desulfurization chemistry at arginine. | Malins, LR., et al. 2013. Chembiochem. 14: 559-63. PMID: 23426906
- Triazine-Based Highly Stable AADD-Type Self-Complementary Quadruple Hydrogen-Bonded Systems Devoid of Prototropy. | Kheria, S., et al. 2017. Chemistry. 23: 783-787. PMID: 27862470
- Synthesis of (+)-(R)-Tiruchanduramine. | Al-Taie, ZS., et al. 2022. Molecules. 27: PMID: 35209136
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N,N′,N′′-Tri-Boc-guanidine, 1 g | sc-301446 | 1 g | $104.00 |
