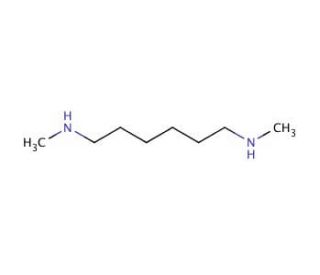
N,N′-Dimethyl-1,6-hexanediamine (CAS 13093-04-4)
QUICK LINKS
N,N′-Dimethyl-1,6-hexanediamine (DMHD) is an organic compound classified as an aliphatic diamine. This colorless, volatile liquid emits a strong ammonia-like odor. With its versatility, N,N′-Dimethyl-1,6-hexanediamine finds widespread applications in the fields of chemistry and biochemistry. In chemistry and biochemistry, N,N′-Dimethyl-1,6-hexanediamine serves as a valuable reagent in various assays. It has proven useful as a substrate in determining enzyme activities, including aminotransferases, amidases, and carboxypeptidases. Additionally, researchers employ it to detect specific proteins and study protein-protein interactions. Moreover, N,N′-Dimethyl-1,6-hexanediamine plays a role in the synthesis of polymers, surfactants, and catalysts. Its involvement in these processes enhances its significance in diverse industrial applications. Though not completely understood, the mechanism of action of N,N′-Dimethyl-1,6-hexanediamine involves acting as a catalyst in the Hofmann-Löffler reaction, a condensation reaction. It is also believed to function as a chelating agent, promoting the stabilization of reaction products. N,N′-Dimethyl-1,6-hexanediamine, with its various applications and roles, holds significant promise in advancing both scientific research and industrial processes.
N,N′-Dimethyl-1,6-hexanediamine (CAS 13093-04-4) References
- Degradable poly(amino alcohol esters) as potential DNA vectors with low cytotoxicity. | Jon, S., et al. 2003. Biomacromolecules. 4: 1759-62. PMID: 14606906
- Fluorophore targeting to cellular proteins via enzyme-mediated azide ligation and strain-promoted cycloaddition. | Yao, JZ., et al. 2012. J Am Chem Soc. 134: 3720-8. PMID: 22239252
- Synthesis of the first poly(diaminosulfide)s and an investigation of their applications as drug delivery vehicles. | Yoo, J., et al. 2012. Macromolecules. 45: 688-697. PMID: 22347726
- New class of biodegradable polymers formed from reactions of an inorganic functional group. | Yoo, J., et al. 2012. Macromolecules. 45: 2292-2300. PMID: 22454554
- Nitric oxide release from polydimethylsiloxane-based polyurethanes. | Nguyen, EB., et al. 2014. J Appl Biomater Funct Mater. 12: 172-82. PMID: 24744231
- Synthesis and Interfacial Activity of Novel Heterogemini Sulfobetaines in Aqueous Solution. | Kwaśniewska, D., et al. 2015. J Surfactants Deterg. 18: 477-486. PMID: 25814830
- Recent advances in thromboresistant and antimicrobial polymers for biomedical applications: just say yes to nitric oxide (NO). | Wo, Y., et al. 2016. Biomater Sci. 4: 1161-83. PMID: 27226170
- Advances in Organic and Organic-Inorganic Hybrid Polymeric Supports for Catalytic Applications. | Salvo, AM., et al. 2016. Molecules. 21: PMID: 27689980
- Multimeric Rhodamine Dye-Induced Aggregation of Silver Nanoparticles for Surface-Enhanced Raman Scattering. | Bartolowits, MD., et al. 2019. ACS Omega. 4: 140-145. PMID: 30729221
- Semi-Crystalline Hydrophobic Polyamidoamines: A New Family of Technological Materials? | Marcioni, M., et al. 2021. Polymers (Basel). 13: PMID: 33806055
- Antimicrobial efficacy of some N,N'-dialkyl-N,N'-dimethyl-1,6-hexanediamine dioxides. | Mlynarcík, D., et al. 1979. Folia Microbiol (Praha). 24: 188-90. PMID: 378783
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N,N′-Dimethyl-1,6-hexanediamine, 1 g | sc-228797 | 1 g | $68.00 |
