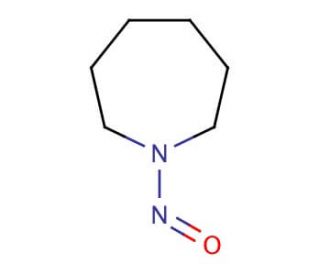
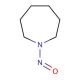
N-Nitrosohexamethyleneimine (CAS 932-83-2)
QUICK LINKS
N-Nitrosohexamethyleneimine is frequently studied in the field of chemical synthesis and environmental science. Researchers investigate its role in the formation of nitrosamines, a class of compounds known for their occurrence in various industrial and natural processes. Studies on N-Nitrosohexamethyleneimine focus on its chemical behavior under different environmental conditions, such as its stability and degradation pathways. This research is essential for understanding how N-Nitrosohexamethyleneimine and similar compounds might impact environmental health. Additionally, N-Nitrosohexamethyleneimine is used in the synthesis of other chemicals, where its reactivity and transformation into different compounds are explored to optimize production processes and reduce potential environmental and health risks.
N-Nitrosohexamethyleneimine (CAS 932-83-2) References
- Attempt to adsorb N-nitrosamines in solution by use of zeolites. | Zhu, JH., et al. 2001. Chemosphere. 44: 949-56. PMID: 11513428
- Free radical chemistry of advanced oxidation process removal of nitrosamines in water. | Landsman, NA., et al. 2007. Environ Sci Technol. 41: 5818-23. PMID: 17874792
- Hierarchical Composites to Reduce N-Nitrosamines in Cigarette Smoke. | Li, YY., et al. 2015. Materials (Basel). 8: 1325-1340. PMID: 28788003
- Strain comparison of systemic N-nitrosohexamethyleneimine carcinogenesis in BALB/c, SENCAR and CD-1 mice. | Strickland, PT., et al. 1988. Cancer Lett. 41: 139-46. PMID: 3401840
- In Silico Drug Screening Analysis against the Overexpression of PGAM1 Gene in Different Cancer Treatments. | Fareed, MM., et al. 2021. Biomed Res Int. 2021: 5515692. PMID: 34195264
- Monitoring a potential carcinogen in pharmaceutical formulations at the low part per billion level. High-performance liquid chromatographic determination of N-nitrosohexamethyleneimine in tolazamide. | Severin, G. 1987. J Chromatogr. 386: 57-63. PMID: 3558617
- Phosphoglycerate mutase 1 that is essential for glycolysis may act as a novel metabolic target for predicating poor prognosis for patients with gastric cancer. | Wei, C., et al. 2022. J Clin Lab Anal. 36: e24718. PMID: 36181311
- Metabolism and mutagenicity of N-nitrosohexamethyleneimine and its hydroxylated derivatives. | Hecker, LI., et al. 1983. Teratog Carcinog Mutagen. 3: 9-17. PMID: 6132461
- In vivo binding of N-nitrosopyrrolidine and N-nitrosohexamethyleneimine to non-purine sites on rat liver DNA. | Ross, AE. and Lawson, TA. 1982. Cancer Lett. 15: 329-34. PMID: 6180827
- Carcinogenesis by N-nitrosohexamethyleneimine in NZO inbred mice. | Goodall, CM. and Lijinsky, W. 1984. Toxicology. 33: 251-9. PMID: 6515660
- Strain and sex differences in N-nitrosohexamethyleneimine carcinogenesis in NZB, NZC, NZO, and NZY mice. | Goodall, CM. and Lijinsky, W. 1984. J Natl Cancer Inst. 73: 1215-8. PMID: 6593491
- Formation of epsilon-hydroxycaproate and epsilon-aminocaproate from N-nitrosohexamethyleneimine: evidence that microsomal alpha-hydroxylation of cyclic nitrosamines may not always involve the insertion of molecular oxygen into the substrate. | Hecker, LI., et al. 1984. Chem Biol Interact. 49: 235-48. PMID: 6722936
- Comparison of the in vitro metabolism of N-nitrosohexamethyleneimine by rat liver and lung microsomal fractions. | Hecker, LI. and McClusky, GA. 1982. Cancer Res. 42: 59-64. PMID: 7053868
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Nitrosohexamethyleneimine, 1 g | sc-215472 | 1 g | $99.00 | |||
N-Nitrosohexamethyleneimine, 5 g | sc-215472A | 5 g | $421.00 |
