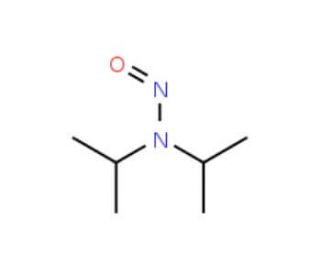
Molecular structure of N-Nitrosodiisopropylamine, CAS Number: 601-77-4
N-Nitrosodiisopropylamine (CAS 601-77-4)
Alternate Names:
N-(1-Methylethyl)-N-nitroso-2-propanamine; 1,1′-Dimethyl-N-nitrosodiethylamine
Application:
N-Nitrosodiisopropylamine is a nitroso compound that shows carcinogenic effects.
CAS Number:
601-77-4
Molecular Weight:
130.19
Molecular Formula:
C6H14N2O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
N-Nitrosodiisopropylamine (NDIPA) is an organic compound widely used in the synthesis of nitro compounds, such as nitroaromatic compounds, nitroalkanes, and nitramines. It reacts with nitric oxide to form nitrosamines, which are known to be carcinogenic and cause cellular damage. It is also used as a reagent for the detection of free radicals. It may act to inhibit the activity of nitric oxide synthase, cyclooxygenase, and superoxide dismutase. Researchers utilize NDIPA to study the role and effects of nitric oxide on the cardiovascular system, nervous system, inflammation, oxidative stress, and the development and progression of cancer.
N-Nitrosodiisopropylamine (CAS 601-77-4) References
- HNO and NO release from a primary amine-based diazeniumdiolate as a function of pH. | Salmon, DJ., et al. 2011. Inorg Chem. 50: 3262-70. PMID: 21405089
- Determination of dimethylamine and nitrite in pharmaceuticals by ion chromatography to assess the likelihood of nitrosamine formation. | Hu, J., et al. 2021. Heliyon. 7: e06179. PMID: 33665410
- Development of a Sensitive Headspace Gas Chromatography-Mass Spectrometry Method for the Simultaneous Determination of Nitrosamines in Losartan Active Pharmaceutical Ingredients. | Wichitnithad, W., et al. 2021. ACS Omega. 6: 11048-11058. PMID: 34056258
- Comparison of EI-GC-MS/MS, APCI-LC-MS/MS, and ESI-LC-MS/MS for the Simultaneous Analysis of Nine Nitrosamines Eluted from Synthetic Resins into Artificial Saliva and Health Risk Assessment. | Kim, H., et al. 2021. Toxics. 9: PMID: 34678926
- Developing Structure-Activity Relationships for N-Nitrosamine Activity. | Cross, KP. and Ponting, DJ. 2021. Comput Toxicol. 20: PMID: 34901581
- A Full Evaporation Static Headspace Gas Chromatography Method with Nitrogen Phosphorous Detection for Ultrasensitive Analysis of Semi-volatile Nitrosamines in Pharmaceutical Products. | Zheng, J., et al. 2022. AAPS J. 24: 23. PMID: 34993666
- Absolute Quantitation of N-Nitrosamines by Coulometric Mass Spectrometry without Using Standards. | Wang, Q., et al. 2022. J Am Soc Mass Spectrom. 33: 875-884. PMID: 35446584
- A substructure-based screening approach to uncover N-nitrosamines in drug substances. | Kao, YT., et al. 2022. J Food Drug Anal. 30: 150-162. PMID: 35647726
- A multi-analyte LC-MS/MS method for screening and quantification of nitrosamines in sartans. | Chang, SH., et al. 2020. J Food Drug Anal. 28: 292-301. PMID: 35696119
- Collaborative reliance in medicine safety and quality regulation: Investigation of experiences in handling N-nitrosamine impurities among ZaZiBoNa participating countries. | Shabangu, PP., et al. 2022. Front Med (Lausanne). 9: 975032. PMID: 36160156
- Determination of Genotoxic Impurity N-Nitroso-N-methyl-4-aminobutyric Acid in Four Sartan Substances through Using Liquid Chromatography-Tandem Mass Spectrometry. | Xie, B., et al. 2022. Molecules. 27: PMID: 36364326
- Risk assessment of N-nitrosamines in food. | , ., et al. 2023. EFSA J. 21: e07884. PMID: 36999063
- Photochemistry of N-nitrosamines in neutral media. | Michejda, CJ. and Rydstrom, T. 1984. IARC Sci Publ. 365-9. PMID: 6533027
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Nitrosodiisopropylamine, 10 mg | sc-212261 | 10 mg | $338.00 | |||
N-Nitrosodiisopropylamine, 25 mg | sc-212261A | 25 mg | $714.00 |
