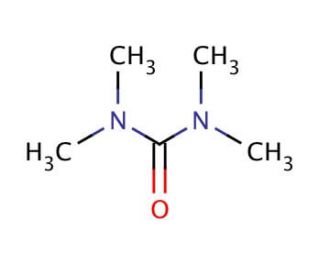
N,N,N′,N′-Tetramethylurea (CAS 632-22-4)
QUICK LINKS
N,N,N′,N′-Tetramethylurea (TMU) is a specialized organic compound notable for its utility in synthetic chemistry, particularly as a high-boiling solvent and in the facilitation of various chemical reactions. Its unique chemical properties, including its ability to dissolve a wide range of compounds, make it an invaluable tool in the synthesis and purification of complex molecules. N,N,N′,N′-Tetramethylurea′s effectiveness as a solvent is attributed to its polar nature, which allows it to engage in hydrogen bonding with solute molecules, thereby enhancing solubility and reaction rates. This characteristic is leveraged in research laboratories to drive reactions that require a stable, non-reactive medium, enabling precise control over reaction conditions. Furthermore, N,N,N′,N′-Tetramethylurea has been employed in the study of polymers and biomolecules, where its solvent properties facilitate the exploration of molecular structures and interactions. Its role extends into the realm of catalysis, where it acts as a medium that can influence the outcome of catalytic processes, thereby providing researchers with the ability to tailor reactions towards desired products. Through these diverse applications, N,N,N′,N′-Tetramethylurea contributes significantly to advancements in chemical synthesis and analytical methods, underscoring its importance in the progress of scientific research and development.
N,N,N′,N′-Tetramethylurea (CAS 632-22-4) References
- An easy access to halide ion-catalytic alpha-glycosylation using carbon tetrabromide and triphenylphosphine as multifunctional reagents. | Shingu, Y., et al. 2003. Org Biomol Chem. 1: 2518-21. PMID: 12956070
- N, N, N′, N′-tetramethylurea (TMU) adducts of the lanthanide isothiocyanates | M. Perrier, G. Vicentini. 1973. Journal of Inorganic and Nuclear Chemistry. 35: 555-559.
- Enthalpies of solution of N,N,N′,N′-tetramethylurea in amides, dimethylsulphoxide, and acetone at 298.15 K | Evgeniy V. Ivanov, Valeriy I. Smirnov. 2008. The Journal of Chemical Thermodynamics. 40: 1342-1345.
- Vibrational Spectroscopic and Theoretical Studies of Urea Derivatives with Biochemical Interest: N,N′-Dimethylurea, N,N,N′,N′-Tetramethylurea, and N,N′-Dimethylpropyleneurea | János Mink, László Hajba, Imre Pápai, Judith Mihály, Csaba Neméth, Mikhail Yu. Skripkin & Magnus Sandström. 2010. Applied Spectroscopy Reviews. 45: 274-326.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N,N,N′,N′-Tetramethylurea, 5 g | sc-250531 | 5 g | $20.00 |
