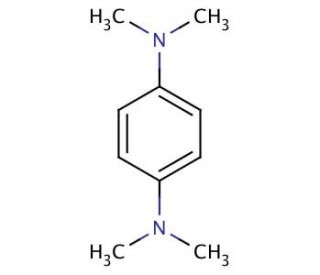
N,N,N′,N′-Tetramethyl-p-phenylenediamine (CAS 100-22-1)
QUICK LINKS
N,N,N′,N′-Tetramethyl-p-phenylenediamine (TMPDA) is an organic compound derived from p-phenylenediamine (PPD), a widely utilized chemical in various product manufacturing processes. N,N,N′,N′-Tetramethyl-p-phenylenediamine, being a versatile compound, finds a multitude of applications in scientific research. N,N,N′,N′-Tetramethyl-p-phenylenediamine has extensive applications in scientific research, encompassing the synthesis of polymers and dyes. It serves as a reagent for metal detection. Moreover, N,N,N′,N′-Tetramethyl-p-phenylenediamine contributes to the synthesis of nanomaterials, including carbon nanotubes. In organic reactions, N,N,N′,N′-Tetramethyl-p-phenylenediamine functions as a catalyst, facilitating the formation of covalent bonds between molecules. This catalytic property enables the creation of more intricate and complex molecules. Additionally, N,N,N′,N′-Tetramethyl-p-phenylenediamine possesses the ability to activate specific chemical reactions, such as the oxidation of organic molecules.
N,N,N′,N′-Tetramethyl-p-phenylenediamine (CAS 100-22-1) References
- Quantitative determination by ESR of the arylaminyl free radical during the reaction of N,N,N',N'-tetramethyl-p-phenylenediamine with oxyhemoglobin. | Störle, C., et al. 1992. Free Radic Res Commun. 17: 133-41. PMID: 1334923
- N,N,N',N'-tetramethyl-p-phenylenediamine initiates the appearance of a well-resolved I peak in the kinetics of chlorophyll fluorescence rise in isolated thylakoids. | Bukhov, NG., et al. 2003. Biochim Biophys Acta. 1607: 91-6. PMID: 14670599
- Oxidation of Manganese and Iron by Leptothrix discophora: Use of N,N,N',N'-Tetramethyl-p-Phenylenediamine as an Indicator of Metal Oxidation. | de Vrind-de Jong, EW., et al. 1990. Appl Environ Microbiol. 56: 3458-62. PMID: 16348351
- Formation and reactions of the Wurster's blue radical cation during the reaction of N,N,N',N'-tetramethyl-p-phenylenediamine with oxyhemoglobin. | Störle, C. and Eyer, P. 1991. Chem Biol Interact. 78: 321-31. PMID: 1649011
- Interaction of N,N,N',N'-tetramethyl-p-phenylenediamine with photosystem II as revealed by thermoluminescence: reduction of the higher oxidation states of the Mn cluster and displacement of plastoquinone from the Q(B) niche. | Gauthier, A., et al. 2006. Biochim Biophys Acta. 1757: 1547-56. PMID: 17064657
- Using N,N,N',N'-tetramethyl-p-phenylenediamine (TMPD) to assay cyclooxygenase activity in vitro. | Petrovic, N. and Murray, M. 2010. Methods Mol Biol. 594: 129-40. PMID: 20072914
- Optical spectra and kinetics of reactions of prostaglandin H synthase: effects of the substrates 13-hydroperoxyoctadeca-9,11-dienoic acid, arachidonic acid, N,N,N',N'-tetramethyl-p-phenylenediamine, and phenol and of the nonsteroidal anti-inflammatory drugs aspirin, indomethacin, phenylbutazone, and bromfenac. | MacDonald, ID., et al. 1989. Arch Biochem Biophys. 272: 194-202. PMID: 2500061
- Janus Reversal and Coulomb Blockade in Ferrocene-Perylenebisimide and N,N,N',N'-Tetramethyl-para-phenylenediamine-Perylenebisimide D-σ-A Rectifiers. | Johnson, MS., et al. 2016. Langmuir. 32: 6851-9. PMID: 27238389
- Generation of superoxide radical, hydrogen peroxide and hydroxyl radical during the autoxidation of N,N,N',N'-tetramethyl-p-phenylenediamine. | Munday, R. 1988. Chem Biol Interact. 65: 133-43. PMID: 2835187
- Robust non-covalent and covalent anchored N,N,N',N'-tetramethyl-p-phenylenediamine derivative on electrode surface via spontaneous physical immobilization and in situ generated aryldiazonium ion electro-grafting: implication for on-surface chemistry and electro-catalytic determinations. | Taleghan Ghafari, M., et al. 2020. Analyst. 145: 596-606. PMID: 31777872
- Near-Infrared Light Triggered a High Temperature Utilizing Donor-Acceptor Cocrystals. | Chen, W., et al. 2022. J Phys Chem Lett. 13: 6571-6579. PMID: 35833756
- Autoxidation of reduced pyridine coenzymes and of their models promoted by N,N,N',N'-tetramethyl-p-phenylenediamine. | Bechara, EJ. and Cilento, G. 1971. Biochemistry. 10: 1831-6. PMID: 4327493
- Tetramethyl-p-phenylenediamine oxidase of Pseudomonas aeruginosa. | Yang, T. 1982. Eur J Biochem. 121: 335-41. PMID: 6800785
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N,N,N′,N′-Tetramethyl-p-phenylenediamine, 5 g | sc-255376 | 5 g | $94.00 |
