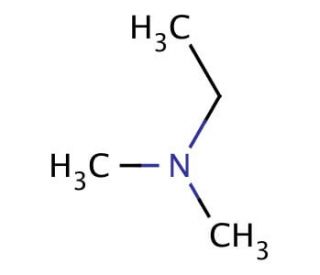
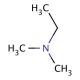
N,N-Dimethylethylamine (CAS 598-56-1)
QUICK LINKS
N,N-Dimethylethylamine functions as a catalyst in certain chemical reactions, facilitating the formation of specific products by lowering the activation energy required for the reaction to occur. It acts as a nucleophilic base, participating in reactions by accepting a proton from an acid and forming a new chemical bond. N,N-Dimethylethylamine can also serve as a ligand in coordination chemistry, binding to metal ions to form stable complexes. In this capacity, it may influence the reactivity and selectivity of metal-catalyzed reactions. N,N-Dimethylethylamine′s mechanism of action involves interacting with other molecules through its amine functional group, enabling it to participate in various chemical transformations. Its ability to donate or accept electrons may be a versatile reagent in chemical synthesis, contributing to the formation of new compounds through its involvement in diverse chemical processes.
N,N-Dimethylethylamine (CAS 598-56-1) References
- Sodium Diisopropylamide in N,N-Dimethylethylamine: Reactivity, Selectivity, and Synthetic Utility. | Ma, Y., et al. 2016. J Org Chem. 81: 11312-11315. PMID: 27768310
- Sodium Diisopropylamide-Mediated Dehydrohalogenations: Influence of Primary- and Secondary-Shell Solvation. | Ma, Y., et al. 2019. J Org Chem. 84: 10860-10869. PMID: 31436099
- Titanium-Catalyzed Intermolecular Hydroaminoalkylation of Alkenes with Tertiary Amines. | Geik, D., et al. 2021. Angew Chem Int Ed Engl. 60: 9936-9940. PMID: 33621392
- Lignin-Based Polyols with Controlled Microstructure by Cationic Ring Opening Polymerization. | Perez-Arce, J., et al. 2021. Polymers (Basel). 13: PMID: 33671706
- Carbonic anhydrase activation profile of indole-based derivatives. | Barresi, E., et al. 2021. J Enzyme Inhib Med Chem. 36: 1783-1797. PMID: 34340630
- Conductivity and Stability Properties of Anion Exchange Membranes: Cation Effect and Backbone Effect. | Han, J., et al. 2021. ChemSusChem. 14: 5021-5031. PMID: 34498428
- Organic amine mediated cleavage of Caromatic-Cα bonds in lignin and its platform molecules. | Xin, Y., et al. 2021. Chem Sci. 12: 15110-15115. PMID: 34909152
- Stereoselective Synthesis of Tertiary Allylic Amines by Titanium-Catalyzed Hydroaminoalkylation of Alkynes with Tertiary Amines. | Kaper, T., et al. 2022. Chemistry. 28: e202103931. PMID: 34936144
- Covalent Attachment of Aggregation-Induced Emission Molecules to the Surface of Ultrasmall Gold Nanoparticles to Enhance Cell Penetration. | Klein, K., et al. 2022. Molecules. 27: PMID: 35335152
- ZnCl2 Mediated Synthesis of InAs Nanocrystals with Aminoarsine. | Zhu, D., et al. 2022. J Am Chem Soc. 144: 10515-10523. PMID: 35648676
- Sodium Isopropyl(trimethylsilyl)amide: A Stable and Highly Soluble Lithium Diisopropylamide Mimic. | Ma, Y., et al. 2022. J Org Chem. 87: 14223-14229. PMID: 36282953
- Near-Infrared Light-Emitting Diodes Based on RoHS-Compliant InAs/ZnSe Colloidal Quantum Dots. | De Franco, M., et al. 2022. ACS Energy Lett. 7: 3788-3790. PMID: 36398094
- Indium arsenide quantum dots: an alternative to lead-based infrared emitting nanomaterials. | Bahmani Jalali, H., et al. 2022. Chem Soc Rev. 51: 9861-9881. PMID: 36408788
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N,N-Dimethylethylamine, 100 g | sc-250519 | 100 g | $62.00 |
