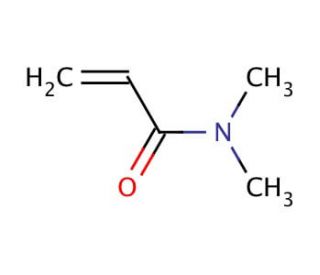
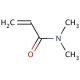
N,N-Dimethylacrylamide (CAS 2680-03-7)
QUICK LINKS
N,N-Dimethylacrylamide functions as a monomer in the synthesis of polymers and copolymers. It plays a role in the formation of poly(N,N-dimethylacrylamide) and other related polymers through polymerization reactions. N,N-Dimethylacrylamide′s mechanism of action involves its ability to undergo radical polymerization, leading to the formation of long chains of repeating units. These polymers can exhibit various properties such as high water solubility, biocompatibility, and responsiveness to external stimuli. N,N-Dimethylacrylamide′s molecular structure allows it to participate in crosslinking reactions, contributing to the development of hydrogels and other polymeric materials with tunable properties. Its functional role in experimental applications involves its use as a building block for the creation of advanced materials with potential applications in various fields.
N,N-Dimethylacrylamide (CAS 2680-03-7) References
- Contamination of 0.2-micrometer infusion filters by N,N-dimethylacrylamide. | Gasch, J., et al. 2010. J Crit Care. 25: 172.e9-14. PMID: 19682842
- Polymer-coated nanoparticles by adsorption of hydrophobically modified poly(N,N-dimethylacrylamide). | Marcelo, G., et al. 2013. J Phys Chem B. 117: 3416-27. PMID: 23496413
- Studies on graft copolymerization of gellan gum with N,N-dimethylacrylamide by the redox system. | Pandey, VS., et al. 2014. Int J Biol Macromol. 70: 108-15. PMID: 24984020
- Influence of cyclodextrin on the UCST- and LCST-behavior of poly(2-methacrylamido-caprolactam)-co-(N,N-dimethylacrylamide). | Burkhart, A. and Ritter, H. 2014. Beilstein J Org Chem. 10: 1951-8. PMID: 25246954
- Temperature and Recognition Dual Responsive Poly(N-Isopropylacrylamide) and Poly(N,N-Dimethylacrylamide) with Adamantyl Side Group. | Dong, Q., et al. 2018. Materials (Basel). 11: PMID: 29565307
- Formation of N, N-Dimethylacrylamide by a Multicenter Hydrocarbamoylation of C2H2 with N, N-Dimethylformamide Activated by Ru5(μ5-C)(CO)15. | Adams, RD. and Tedder, JD. 2018. Inorg Chem. 57: 5707-5710. PMID: 29708739
- N,N-dimethylacrylamide-A new sensitizer in the FreeStyle Libre glucose sensor. | Mowitz, M., et al. 2019. Contact Dermatitis. 81: 27-31. PMID: 30773644
- Formose Reaction Controlled by a Copolymer of N,N-Dimethylacrylamide and 4-Vinylphenylboronic Acid. | Michitaka, T., et al. 2017. Polymers (Basel). 9: PMID: 30965856
- Water-Stable Plasma-Polymerized N,N-Dimethylacrylamide Coatings to Control Cellular Adhesion. | Egghe, T., et al. 2020. ACS Appl Mater Interfaces. 12: 2116-2128. PMID: 31834769
- Synthesis and Heavy-Metal Sorption Studies of N,N-Dimethylacrylamide-Based Hydrogels. | Akhmetzhan, A., et al. 2021. Polymers (Basel). 13: PMID: 34577985
- A Short Review on the N,N-Dimethylacrylamide-Based Hydrogels. | Akhmetzhan, A., et al. 2021. Gels. 7: PMID: 34940294
- Poly(N,N-dimethylacrylamide-octadecyl acrylate)-clay hydrogels with high mechanical properties and shape memory ability. | Wang, F., et al. 2018. RSC Adv. 8: 16773-16780. PMID: 35540536
- Optimization of Functional Group Concentration of N, N-Dimethylacrylamide-based Polymeric Coatings and Probe Immobilization for DNA and Protein Microarray Applications. | Sola, L., et al. 2023. Micromachines (Basel). 14: PMID: 36838001
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N,N-Dimethylacrylamide, 5 ml | sc-255370 | 5 ml | $44.00 | |||
N,N-Dimethylacrylamide, 100 ml | sc-255370A | 100 ml | $52.00 |
