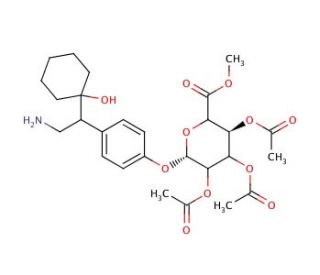
N,N-Didesmethyl-O-desmethyl rac-Venlafaxine 2,3,4-Tri-O-acetyl-β-D-glucuronide Methyl Ester
QUICK LINKS
N,N-Didesmethyl-O-desmethyl rac-Venlafaxine 2,3,4-Tri-O-acetyl-β-D-glucuronide Methyl Ester is an intermediate for the synthesis of N,N-Didesmethyl-O-desmethyl-(rac-Venlafaxine) Glucuronide (sc-219275). N,N-Didesmethyl-O-desmethyl rac-Venlafaxine 2,3,4-Tri-O-acetyl-β-D-glucuronide Methyl Ester is also a derivative of Venlafaxine (sc-201102). Venlafaxine is a derivative of phenylethylamine which is reported to facilitate neurotransmission within the central nervous system via blocking the presynaptic reuptake of neuroamines such as serotonin (5-hydroxytryptamine; 5-HT) and noradrenaline (norepinephrine). Velanfaxine is also reported to be a weak inhibitor of dopamine reuptake. In vitro studies indicate that Venlafaxine does not demonstrate significant activity for muscarinic, histaminergic or α-1 adrenergic receptors. The metabolism of venlafaxine is reported to occur by cytochrome P450 (CYP) enzyme CYP2D6 yielding O-desmethylvenlafaxine. A lesser metabolite, N-desmethylvenlafaxine is produced by CYP3A4.
N,N-Didesmethyl-O-desmethyl rac-Venlafaxine 2,3,4-Tri-O-acetyl-β-D-glucuronide Methyl Ester References
- Comparative affinity of duloxetine and venlafaxine for serotonin and norepinephrine transporters in vitro and in vivo, human serotonin receptor subtypes, and other neuronal receptors. | Bymaster, FP., et al. 2001. Neuropsychopharmacology. 25: 871-80. PMID: 11750180
- SNRIs: their pharmacology, clinical efficacy, and tolerability in comparison with other classes of antidepressants. | Stahl, SM., et al. 2005. CNS Spectr. 10: 732-47. PMID: 16142213
- Effects of different doses of venlafaxine on serotonin and norepinephrine reuptake in healthy volunteers. | Blier, P., et al. 2007. Int J Neuropsychopharmacol. 10: 41-50. PMID: 16690005
- CYP2D6 polymorphism and clinical effect of the antidepressant venlafaxine. | Shams, ME., et al. 2006. J Clin Pharm Ther. 31: 493-502. PMID: 16958828
- In vivo effect of venlafaxine on locus coeruleus neurons: role of opioid, alpha(2)-adrenergic, and 5-hydroxytryptamine(1A) receptors. | Berrocoso, E. and Mico, JA. 2007. J Pharmacol Exp Ther. 322: 101-7. PMID: 17431134
- Venlafaxine. A review of its pharmacology and therapeutic potential in depression. | Holliday, SM. and Benfield, P. 1995. Drugs. 49: 280-94. PMID: 7729333
- Venlafaxine: a new antidepressant drug. | Holdcroft, C. 1994. Nurse Pract. 19: 21. PMID: 7816367
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N,N-Didesmethyl-O-desmethyl rac-Venlafaxine 2,3,4-Tri-O-acetyl-β-D-glucuronide Methyl Ester, 1 mg | sc-224152 | 1 mg | $490.00 |
