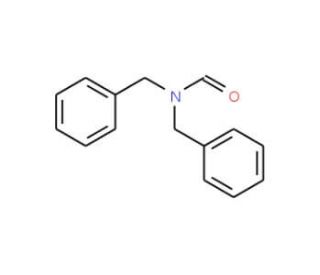
N,N-Dibenzylformamide (CAS 5464-77-7)
QUICK LINKS
N,N-Dibenzylformamide is a chemical compound that finds application in various research disciplines, primarily within organic chemistry. It serves as a useful intermediate in the synthesis of other chemical entities, particularly those that require the introduction of benzyl protecting groups. The formamide moiety in N,N-Dibenzylformamide is a key functional group that researchers exploit in the study of amide bond formation and cleavage reactions. This compound is also of interest in the field of catalysis, where it may be used to investigate the efficiency and selectivity of new catalysts, especially in the context of selective N-benzylation. Furthermore, its stability and reactivity make N,N-Dibenzylformamide a candidate for exploring the principles of hydrogen bonding and dipole interactions in supramolecular chemistry.
N,N-Dibenzylformamide (CAS 5464-77-7) References
- Intramolecular and Intermolecular Schmidt Reactions of Alkyl Azides with Aldehydes. | Lee, HL. and Aubé, J. 2007. Tetrahedron. 63: 9007-9015. PMID: 18769536
- Direct thionation and selenation of amides using elemental sulfur and selenium and hydrochlorosilanes in the presence of amines. | Shibahara, F., et al. 2009. Org Lett. 11: 3064-7. PMID: 19534478
- Scalable synthesis of enaminones utilizing Gold's reagents. | Schuppe, AW., et al. 2017. Tetrahedron Asymmetry. 73: 3643-3651. PMID: 31827315
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N,N-Dibenzylformamide, 1 g | sc-212305 | 1 g | $300.00 |
