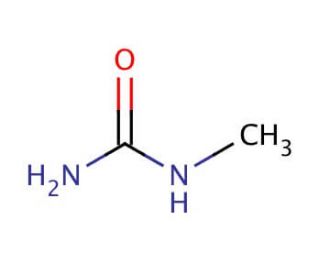
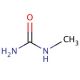
N-Methylurea (CAS 598-50-5)
QUICK LINKS
N-Methylurea is a compound that functions as a reactant in various chemical synthesis processes. It acts as a methylating agent, participating in reactions that involve the transfer of a methyl group to other molecules. N-Methylurea′s mechanism of action involves its ability to donate a methyl group to specific substrates, leading to the formation of new compounds with altered chemical properties. N-Methylurea′s role in these reactions is to facilitate the introduction of methyl groups into target molecules, thereby influencing their structure and reactivity. At the molecular level, N-Methylurea interacts with specific functional groups within the substrates, leading to the modification of their chemical composition. N-Methylurea serves as a reagent for the synthesis of various organic compounds, contributing to the creation of new materials and substances with modified chemical properties.
N-Methylurea (CAS 598-50-5) References
- Three-component reaction between triphenylphosphine, dialkyl acetylenedicarboxylates and urea or N-methylurea. | Yavari, I. and Zabarjad-Shiraz, N. 2006. Mol Divers. 10: 23-7. PMID: 16404526
- Transnitrosation of non-mutagenic N-nitrosoproline forms mutagenic N-nitroso-N-methylurea. | Inami, K., et al. 2015. Bioorg Med Chem. 23: 3297-302. PMID: 25975641
- Tailoring Fe2O3-Al2O3 catalyst structure and activity via hydrothermal synthesis for carbon nanotubes and hydrogen production from polyolefin plastics. | Veksha, A., et al. 2022. Chemosphere. 297: 134148. PMID: 35240158
- Green Synthesis of Thiazolidine-2,4-dione Derivatives and Their Lipoxygenase Inhibition Activity With QSAR and Molecular Docking Studies. | Lončarić, M., et al. 2022. Front Chem. 10: 912822. PMID: 35864866
- Choline Chloride-Based Deep Eutectic Solvents as Green Effective Medium for Quaternization Reactions. | Bušić, V., et al. 2022. Molecules. 27: PMID: 36364264
- Urea Decomposition Mechanism by Dinuclear Nickel Complexes. | Martins, CO., et al. 2023. Molecules. 28: PMID: 36838646
- Highly Efficient Recovery and Recycling of Cobalt from Spent Lithium-Ion Batteries Using an N-Methylurea-Acetamide Nonionic Deep Eutectic Solvent. | Suriyanarayanan, S., et al. 2023. ACS Omega. 8: 6959-6967. PMID: 36844576
- Hydroxyurea pharmacokinetics and precision dosing in low-resource settings. | Smart, LR., et al. 2023. Front Mol Biosci. 10: 1130206. PMID: 37325474
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Methylurea, 100 g | sc-250483 | 100 g | $37.00 |
