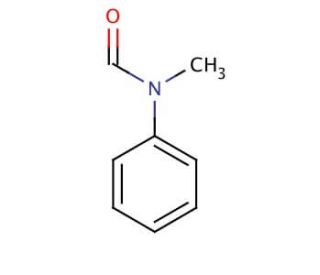
N-Methylformanilide (CAS 93-61-8)
QUICK LINKS
N-Methylformanilide is a solvent used in development applications. It functions as a reaction medium for various chemical reactions, particularly those involving metal-catalyzed processes. Its mechanism of action involves facilitating the dissolution of reactants and the formation of intermediates, allowing for efficient progress of the desired chemical transformations. N-Methylformanilide is known to interact with substrates and catalysts at the molecular level, promoting the desired chemical reactions without directly participating in the reaction itself. N-Methylformanilide′s role in experimental applications involves providing a suitable environment for the synthesis of various compounds, enabling to carry out specific reactions under controlled conditions. Its mechanism of action at the molecular level allows for the manipulation of chemical structures and the production of desired products in a controlled and efficient manner.
N-Methylformanilide (CAS 93-61-8) References
- Selective electrocatalytic oxidation of N-alkyl-N-methylanilines to N-alkylformanilides using nitroxyl radical. | Kashiwagi, Y. and Anzai, J. 2001. Chem Pharm Bull (Tokyo). 49: 324-6. PMID: 11253925
- The unique nucleophilic reactivity of arylaminochlorocarbenes. | Cheng, Y., et al. 2003. Chem Commun (Camb). 90-1. PMID: 12610980
- Sustainable heterogeneous platinum catalyst for direct methylation of secondary amines by carbon dioxide and hydrogen. | Kon, K., et al. 2014. Chemistry. 20: 6264-7. PMID: 24757090
- Direct condensation of functionalized sp(3) carbons with formanilides for enamine synthesis using an in situ generated HMDS amide catalyst. | Taneda, H., et al. 2014. Chem Commun (Camb). 50: 6523-5. PMID: 24820148
- Enhancing the Dyeability of Polyimide Fibers with the Assistance of Swelling Agents. | Shao, D., et al. 2019. Materials (Basel). 12: PMID: 30678312
- Highly selective hydrogenation of amides catalysed by a molybdenum pincer complex: scope and mechanism. | Leischner, T., et al. 2019. Chem Sci. 10: 10566-10576. PMID: 32110342
- Lipophilicity study of different cephalosporins: Computational prediction of minimum inhibitory concentration using salting-out chromatography. | AboulMagd, AM., et al. 2021. J Pharm Biomed Anal. 206: 114358. PMID: 34534866
- Light-induced dissolution and concomitant crystallization of a Keggin-type polyoxometalate mimicking a naturally occurring phenomenon. | Tandekar, K., et al. 2023. Chem Commun (Camb). 59: 3241-3244. PMID: 36815372
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Methylformanilide, 5 g | sc-250477 | 5 g | $23.00 | |||
N-Methylformanilide, 100 g | sc-250477A | 100 g | $27.00 |
