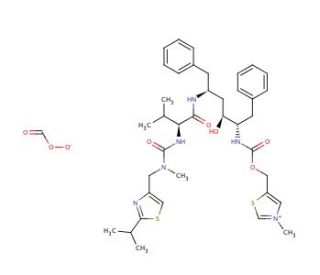
N-Methyl Ritonavir Bicarbonate
QUICK LINKS
N-Methyl Ritonavir Bicarbonate is a modified analog of ritonavir where a methyl group is added to the nitrogen atom, and a bicarbonate functional group is introduced. This chemical modification is intended to study the biochemical and biophysical properties of ritonavir, particularly how these modifications affect its interaction with various enzymes and proteins. The primary mechanism of action for the parent compound, ritonavir, involves the inhibition of HIV-1 protease, an essential enzyme that the virus needs to process its polyprotein precursors into mature proteins, which are necessary for assembling new viral particles. By inhibiting this enzyme, ritonavir prevents the maturation of the virus, leading to the production of non-infectious viral particles. N-Methyl Ritonavir Bicarbonate is used in research to see how the methylation and the addition of a bicarbonate group influence this mechanism. In scientific studies, N-Methyl Ritonavir Bicarbonate is used to dissect the enzyme kinetics and binding dynamics of ritonavir. Researchers utilize this compound to investigate how structural changes impact the binding affinity and inhibition efficiency against HIV-1 protease. The introduction of the methyl and bicarbonate groups can provide insights into the role of specific functional groups in the interaction with the protease and can help to map out the enzyme′s active site more precisely. Additionally, N-Methyl Ritonavir Bicarbonate is used in computational modeling and molecular dynamics simulations. These studies help to visualize and predict how the modified molecule interacts at the atomic level with the protease, offering a deeper understanding of the inhibitory mechanism and how it might be enhanced or altered by such modifications.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Methyl Ritonavir Bicarbonate, 5 mg | sc-219181 | 5 mg | $380.00 |
