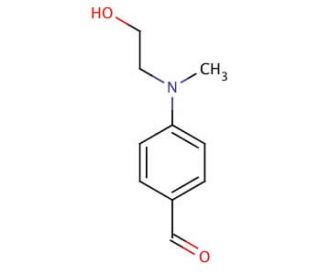
N-Methyl-N-(2-hydroxyethyl)-4-aminobenzaldehyde (CAS 1201-91-8)
QUICK LINKS
N-Methyl-N-(2-hydroxyethyl)-4-aminobenzaldehyde, also known as NMEAB or 4-methoxybenzaldehyde, is an important compound derived from the aromatic aldehyde family. With its unique structure consisting of a benzene ring bearing a methyl group and a hydroxyethyl group attached to the nitrogen atom, N-Methyl-N-(2-hydroxyethyl)-4-aminobenzaldehyde finds extensive applications in scientific research. As a reagent, N-Methyl-N-(2-hydroxyethyl)-4-aminobenzaldehyde plays a role in the synthesis of heterocyclic compounds such as pyridines, indoles, and quinolines. It serves as a starting material for the synthesis of various pharmaceuticals, fragrances, and other valuable products. In the realm of scientific exploration, N-Methyl-N-(2-hydroxyethyl)-4-aminobenzaldehyde has been employed in the synthesis of biologically active compounds, including inhibitors of dihydrofolate reductase, cyclooxygenase-2, and 5-lipoxygenase. N-Methyl-N-(2-hydroxyethyl)-4-aminobenzaldehyde exhibits solubility in both water and organic solvents, adding to its versatility in various experimental setups. The reactivity of N-Methyl-N-(2-hydroxyethyl)-4-aminobenzaldehyde stems from its mechanism of action, which involves the nucleophilic substitution reaction of the methyl group with the nitrogen atom in the benzene ring. This reaction leads to the formation of a new carbon-nitrogen bond, ultimately yielding the desired product.
N-Methyl-N-(2-hydroxyethyl)-4-aminobenzaldehyde (CAS 1201-91-8) References
- Photoswitchable fluorescent dyads incorporating BODIPY and [1,3]oxazine components. | Deniz, E., et al. 2010. J Phys Chem A. 114: 11567-75. PMID: 20939622
- Synthesis and evaluation of chalcone derivatives as inhibitors of neutrophils' chemotaxis, phagocytosis and production of reactive oxygen species. | Bukhari, SN., et al. 2014. Chem Biol Drug Des. 83: 198-206. PMID: 24433224
- Synthesis and In Vitro Evaluation of a Photosensitizer-BODIPY Derivative for Potential Photodynamic Therapy Applications. | Gorbe, M., et al. 2015. Chem Asian J. 10: 2121-5. PMID: 26282179
- Therapeutic Nanosystem Consisting of Singlet-Oxygen-Responsive Prodrug and Photosensitizer Excited by Two-Photon Light. | Lin, Y., et al. 2018. ACS Med Chem Lett. 9: 23-27. PMID: 29348806
- Acceptor Influence on Thiolate Sensing by Hemicyanine Dyes. | Chung, AJ., et al. 2019. J Org Chem. 84: 2261-2268. PMID: 30664354
- Development of a fluorine-18 radiolabelled fluorescent chalcone: evaluated for detecting glycogen. | Allott, L., et al. 2020. EJNMMI Radiopharm Chem. 5: 17. PMID: 32578021
- Synthesis and characterization of dendritic nonlinear optical chromophore containing phenylene attached with bulky alkyl group | , et al. (2007). Macromolecular Research. volume 15:, pages 59–64.
- Off–on BODIPY-based chemosensors for selective detection of Al3+ and Cr3+ versus Fe3+ in aqueous media† | Andrea Barba-Bonac, Laura Calabuigb, Ana M. Costero*ab, Salvador Gilab, Ramón Martínez-Máñez*ac and Félix Sancenónac. 2014,. RSC Adv.,. 4,: 8962-8965.
- Effects of Novel Diarylpentanoid Analogues of Curcumin on Secretory Phospholipase A2, Cyclooxygenases, Lipo-oxygenase, and Microsomal Prostaglandin E Synthase-1 | and Waqas Ahmad, Endang Kumolosasi, Ibrahim Jantan, Syed N. A. Bukhari, Malina Jasamai. June 2014. Chemical Biology & Drug Design. Volume83, Issue6: Pages 670-681.
- Investigation of electronic energy transfer in a BODIPY-decorated calix[4]arene | I Tosi, B Bardi, M Ambrosetti, E Domenichini, A Iagatti… - Dyes and …, 2019 - Elsevier. December 2019,. Dyes and Pigments. Volume 171,: 107652.
- Synthesis and evaluation of novel two-photon fluorescence probes for in vivo imaging of amylin aggregates in the pancreas | H Watanabe, Y Miki, Y Shimizu, H Saji, M Ono - Dyes and Pigments, 2019 - Elsevier. November 2019,. Dyes and Pigments. Volume 170,: 107615.
- Chalcone-based fluorescent chemosensors as new tools for detecting Cu2+ ions | LJ Gomes, T Moreira, L Rodríguez, AJ Moro - Dyes and Pigments, 2022 - Elsevier. January 2022,. Dyes and Pigments. Volume 197,: 109845.
- Synthesis, spectral characterization, chemical reactivity and anticancer behaviors of some novel hydrazone derivatives: Experimental and theoretical insights | R Çakmak, E Başaran, S Kaya, S Erkan -. 5 April 2022,. Journal of Molecular Structure. Volume 1253,: 132224.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Methyl-N-(2-hydroxyethyl)-4-aminobenzaldehyde, 25 g | sc-228727 | 25 g | $108.00 |
