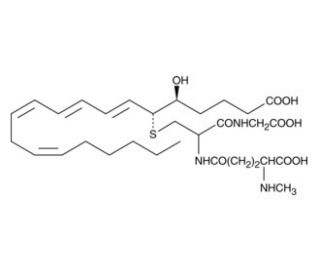
N-methyl Leukotriene C4 (CAS 131391-65-6)
QUICK LINKS
N-methyl Leukotriene C4 is a synthetic analog of leukotriene C4 (LTC4) that is not readily metabolized to LTD4 and LTE4. N-methyl LTC4 has about a 30-fold reduced binding affinity to LTC4 receptor-expressing DMSO-differentiated U937 cell membranes compared to LTC4. LTC4 is the parent cysteinyl leukotriene formed by the LTC4 synthase-catalyzed conjugation of glutathione to LTA4. Its production has been observed in neutrophils, mast cells, and macrophages, as well as by transcellular metabolism in platelets. It may act to be one of the primary constituents of slow-reacting substance of anaphylaxis (SRS-A) and exhibits potent smooth muscle contracting activity. LTC4 is rapidly metabolized to LTD4 and LTE4, making the characterization of LTC4 pharmacology difficult.
N-methyl Leukotriene C,[object Object], (CAS 131391-65-6) References
- Prostaglandins and leukotrienes: advances in eicosanoid biology. | Funk, CD. 2001. Science. 294: 1871-5. PMID: 11729303
- Differential signaling of cysteinyl leukotrienes and a novel cysteinyl leukotriene receptor 2 (CysLT₂) agonist, N-methyl-leukotriene C₄, in calcium reporter and β arrestin assays. | Yan, D., et al. 2011. Mol Pharmacol. 79: 270-8. PMID: 21078884
- Bioconversion of leukotriene D4 by lung dipeptidase. | Campbell, BJ., et al. 1990. Biochim Biophys Acta. 1042: 107-12. PMID: 2153408
- HAMI 3379, a CysLT2 receptor antagonist, attenuates ischemia-like neuronal injury by inhibiting microglial activation. | Zhang, XY., et al. 2013. J Pharmacol Exp Ther. 346: 328-41. PMID: 23750020
- Transcellular metabolism of neutrophil-derived leukotriene A4 by human platelets. A potential cellular source of leukotriene C4. | Maclouf, JA. and Murphy, RC. 1988. J Biol Chem. 263: 174-81. PMID: 2826437
- A basophil-neuronal axis promotes itch. | Wang, F., et al. 2021. Cell. 184: 422-440.e17. PMID: 33450207
- Exosomal miRNA-223-3p as potential biomarkers in patients with cerebral small vessel disease cognitive impairment. | Zhao, W., et al. 2021. Ann Transl Med. 9: 1781. PMID: 35071475
- Formation and actions of leukotrienes. | Piper, PJ. 1984. Physiol Rev. 64: 744-61. PMID: 6324254
- Microsomal glutathione S-transferase is the predominant leukotriene C4 binding site in cellular membranes. | Metters, KM., et al. 1994. J Biol Chem. 269: 12816-23. PMID: 8175695
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-methyl Leukotriene C4, 25 µg | sc-205407 | 25 µg | $285.00 | |||
N-methyl Leukotriene C4, 100 µg | sc-205407A | 100 µg | $1015.00 | |||
N-methyl Leukotriene C4, 1 mg | sc-205407B | 1 mg | $9370.00 |
