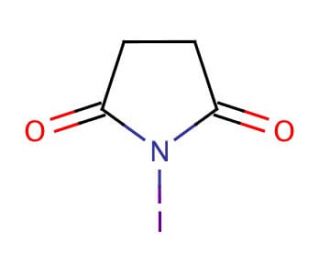
Molecular structure of N-Iodosuccinimide, CAS Number: 516-12-1
N-Iodosuccinimide (CAS 516-12-1)
Application:
N-Iodosuccinimide is a reagent for the oxidimetric titration of sulfur functions, oxidations, and iodinations
CAS Number:
516-12-1
Molecular Weight:
224.98
Molecular Formula:
C4H4INO2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
N-Iodosuccinimide is a reagent for the oxidimetric titration of sulfur functions, oxidations, and iodinations.
N-Iodosuccinimide (CAS 516-12-1) References
- Determination of sulphur functions with N-iodosuccinimide. | Srivastava, A. 1979. Talanta. 26: 917-20. PMID: 18962543
- Umpolung amide synthesis using substoichiometric N-iodosuccinimide (NIS) and oxygen as a terminal oxidant. | Schwieter, KE., et al. 2014. Org Lett. 16: 4714-7. PMID: 25198239
- Highly Regioselective Iodination of Arenes via Iron(III)-Catalyzed Activation of N-Iodosuccinimide. | Racys, DT., et al. 2015. Org Lett. 17: 4782-5. PMID: 26394175
- N-Iodosuccinimide involved one-pot metal-free synthesis of 2-heteroaromatic benzothiazole compounds. | Chu, X., et al. 2017. Org Biomol Chem. 15: 1606-1611. PMID: 28116402
- Preparation of 3-Iodoquinolines from N-Tosyl-2-propynylamines with Diaryliodonium Triflate and N-Iodosuccinimide. | Sasaki, T., et al. 2017. J Org Chem. 82: 11727-11734. PMID: 28718287
- Straightforward S-S Bond Formation via the Oxidation of S-Acetyl by Iodine in the Presence of N-Iodosuccinimide. | Ge, JT., et al. 2017. J Org Chem. 82: 12613-12623. PMID: 29084384
- Trifluoromethylation of Unactivated Alkenes with Me3SiCF3 and N-Iodosuccinimide. | Yang, X. and Tsui, GC. 2019. Org Lett. 21: 1521-1525. PMID: 30777764
- A novel approach for the detection and identification of sulfur mustard using liquid chromatography-electrospray ionization-tandem mass spectrometry based on its selective oxidation to sulfur mustard monoxide with N-iodosuccinimide. | Prihed, H., et al. 2021. J Mass Spectrom. 56: e4721. PMID: 33848030
- Intramolecular C-H Amination of N-Alkylsulfamides by tert-Butyl Hypoiodite or N-Iodosuccinimide. | Kiyokawa, K., et al. 2021. Chemistry. 27: 13971-13976. PMID: 34403187
- A pentacarbomethoxycyclopentadiene (PCCP) organic Brønsted acid catalyzed stereoselective glycosidation of N-pentenyl orthoesters (NPOE) of d-glucose and d-galactose, in conjunction with N-iodosuccinimide. | Aswathy, M., et al. 2022. Carbohydr Res. 522: 108684. PMID: 36193594
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Iodosuccinimide, 5 g | sc-255356 | 5 g | $48.00 | |||
N-Iodosuccinimide, 25 g | sc-255356A | 25 g | $179.00 |
