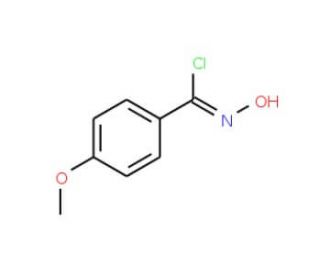
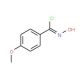
N-Hydroxy-4-methoxybenzenecarboximidoyl chloride (CAS 38435-51-7)
QUICK LINKS
N-Hydroxy-4-methoxybenzenecarboximidoyl chloride is a compound that functions as a reagent in organic synthesis. It acts as a versatile intermediate in the preparation of various organic compounds, particularly in the formation of amides, esters, and other functional groups. The mechanism of action involves its ability to undergo nucleophilic substitution reactions, allowing for the introduction of the carboximidoyl chloride group into organic molecules. N-Hydroxy-4-Methoxybenzenecarboximidoyl Chloride participates in reactions that lead to the formation of new carbon-nitrogen bonds, contributing to the synthesis of complex organic structures. Its role in the modification of organic molecules may be a useful for the development of novel compounds with potential applications in various fields of research.
N-Hydroxy-4-methoxybenzenecarboximidoyl chloride (CAS 38435-51-7) References
- Synthesis, characterization and biological evaluation of some novel 17-isoxazoles in the estrone series. | Kovács, D., et al. 2012. Steroids. 77: 1075-85. PMID: 22613036
- Synthesis of novel steroidal 16-spiroisoxazolines by 1,3-dipolar cycloaddition, and an evaluation of their antiproliferative activities in vitro. | Frank, É., et al. 2014. Mol Divers. 18: 521-34. PMID: 24691697
- Regio- and stereoselective access to novel ring-condensed steroidal isoxazolines. | Mótyán, G., et al. 2014. Steroids. 87: 76-85. PMID: 24928722
- Regio-and stereoselective synthesis of pregnane-fused isoxazolines by nitril-oxide/alkene 1, 3-dipolar cycloaddition and an evaluation of their cell-growth inhibitory effect in vitro | Mótyán, G., Baji, Á., Zupkó, I., & Frank, É. 2016. Journal of Molecular Structure. 1110: 143-149.
- Microwave-assisted synthesis of (3,5-disubstituted isoxazole)-linked benzimidazolone derivatives: DFT calculations and biological activities | Ibrahim, S., Ghabi, A., Amiri, N., Mtiraoui, H., Hajji, M., Bel-Hadj-Tahar, R., & Msaddek, M. 2021. Monatshefte für Chemie-Chemical Monthly. 152(5): 523-535.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Hydroxy-4-methoxybenzenecarboximidoyl chloride, 500 mg | sc-301353 | 500 mg | $149.00 | |||
N-Hydroxy-4-methoxybenzenecarboximidoyl chloride, 1 g | sc-301353A | 1 g | $207.00 |
