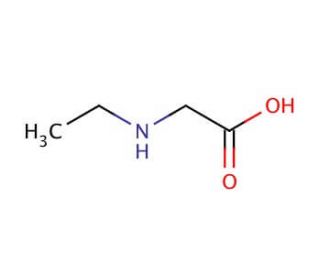
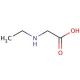
N-Ethylglycine (CAS 627-01-0)
QUICK LINKS
N-ethylglycine (NEG), or ethylglycine, is an amino acid derivative extensively employed in diverse scientific research endeavors. This small molecule bears structural similarity to glycine and undergoes a two-step synthesis from glycine and ethyl bromide. Its versatile nature allows it to be utilized in various scientific experiments, unveiling a range of biochemical and physiological effects. N-ethylglycine finds application in multiple scientific research domains, including investigations into enzyme kinetics, protein structure and function, and drug metabolism. It serves as useful for studying the impacts of drugs on the nervous system and exploring the mechanisms of drug action. Functionally, N-ethylglycine is believed to act as an enzyme inhibitor by binding to the active site of the enzyme, impeding its proper functioning. Additionally, it is considered a modulator of signal transduction pathways, exerting its effects by binding to proteins involved in signal transduction. The multifaceted utility of N-ethylglycine in scientific research enables the elucidation of enzyme behavior, protein function, drug effects, and signal transduction mechanisms. Through its inhibitory properties and modulation of signal transduction, N-ethylglycine contributes to the understanding of various biological processes and pathways.
N-Ethylglycine (CAS 627-01-0) References
- Monomeric sarcosine oxidase: 2. Kinetic studies with sarcosine, alternate substrates, and a substrate analogue. | Wagner, MA. and Jorns, MS. 2000. Biochemistry. 39: 8825-9. PMID: 10913293
- Glycine oxidase from Bacillus subtilis. Characterization of a new flavoprotein. | Job, V., et al. 2002. J Biol Chem. 277: 6985-93. PMID: 11744710
- Identification of N-ethylglycine in urine of cancer patients with metastatic bone disease. | Tsuruta, Y., et al. 2007. Clin Chim Acta. 376: 226-8. PMID: 16962088
- Determination of N-ethylglycine in urine of cancer patients with metastatic bone disease by HPLC using 4-(5,6-dimethoxy-2-phthalimidinyl)-2-methoxyphenylsulfonyl chloride as a fluorescent labeling reagent. | Tsuruta, Y., et al. 2008. Anal Sci. 24: 1629-31. PMID: 19075476
- Fully automated solid-phase microextraction-fast gas chromatography-mass spectrometry method using a new ionic liquid column for high-throughput analysis of sarcosine and N-ethylglycine in human urine and urinary sediments. | Bianchi, F., et al. 2011. Anal Chim Acta. 707: 197-203. PMID: 22027139
- Lidocaine metabolites inhibit glycine transporter 1: a novel mechanism for the analgesic action of systemic lidocaine? | Werdehausen, R., et al. 2012. Anesthesiology. 116: 147-58. PMID: 22133759
- Lidocaine metabolite and creatinine measurements in the Ektachem 700: steps to minimize its impact on patient care. | Sena, SF., et al. 1988. Clin Chem. 34: 2144-8. PMID: 2458864
- Endohedral metal-induced regioselective formation of bis-Prato adduct of Y3N@I(h)-C80 and Gd3N@I(h)-C80. | Aroua, S., et al. 2015. J Am Chem Soc. 137: 58-61. PMID: 25549139
- The lidocaine metabolite N-ethylglycine has antinociceptive effects in experimental inflammatory and neuropathic pain. | Werdehausen, R., et al. 2015. Pain. 156: 1647-1659. PMID: 25932687
- Definitive liquid-chromatographic demonstration that N-ethylglycine is the metabolite of lidocaine that interferes in the Kodak sarcosine oxidase-coupled method for creatinine. | Roberts, RT., et al. 1988. Clin Chem. 34: 2569-72. PMID: 3197304
- Regioselective Synthesis and Characterization of Tris- and Tetra-Prato Adducts of M3N@C80 (M = Y, Gd). | Semivrazhskaya, O., et al. 2020. J Am Chem Soc. 142: 12954-12965. PMID: 32586092
- Tetrabutylammonium Bromide (TBAB) Catalyzed Synthesis of Bioactive Heterocycles. | Banik, BK., et al. 2020. Molecules. 25: PMID: 33327504
- Formation of amino acids on heating glycine with alumina. | Ivanov, CP. and Slavcheva, NN. 1977. Orig Life. 8: 13-9. PMID: 896188
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Ethylglycine, 1 g | sc-236040 | 1 g | $204.00 |
