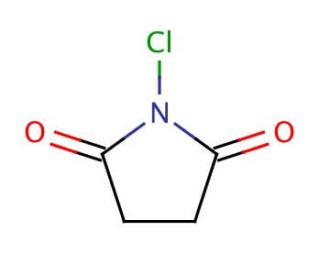
N-Chlorosuccinimide (CAS 128-09-6)
QUICK LINKS
N-Chlorosuccinimide is a regioselective chlorination and oxidizing reagent. N-Chlorosuccinimide (NCS) is a widely used chemical reagent in the fields of synthetic organic chemistry and biochemistry. It plays a role as an intermediate in the synthesis of diverse compounds, including dyes and agrochemicals. Moreover, N-Chlorosuccinimide is highly valuable for creating N-chloro compounds, which find applications in various areas. Beyond its significance in scientific research, N-Chlorosuccinimide holds versatility as a reagent in a multitude of synthetic reactions. When N-Chlorosuccinimide interacts with organic substrates, it forms N-chloro compounds through a reaction initiated by the generation of a chlorinating agent. This chlorinating agent originates from the reaction between N-Chlorosuccinimide and a base, leading to the formation of the desired N-chloro compound. N-Chlorosuccinimide stands as a reagent with wide-ranging utility in both synthetic organic chemistry and biochemistry. Its diverse applications encompass the synthesis of various compounds and its role in the formation of essential N-chloro compounds, making it an indispensable tool for scientific exploration and innovation.
N-Chlorosuccinimide (CAS 128-09-6) References
- Synthesis of sulfonimidamides from sulfinamides by oxidation with N-chlorosuccinimide. | Mancheño, OG. and Bolm, C. 2007. Beilstein J Org Chem. 3: 25. PMID: 17894869
- Flow injection chemiluminescence determination of sulfide by oxidation with N-bromosuccinimide and N-chlorosuccinimide. | Safavi, A. and Karimi, MA. 2002. Talanta. 57: 491-500. PMID: 18968648
- N-Chlorosuccinimide, an efficient reagent for on-resin disulfide formation in solid-phase peptide synthesis. | Postma, TM. and Albericio, F. 2013. Org Lett. 15: 616-9. PMID: 23320397
- Flow-injection chemiluminescence determination of olanzapine using N-chlorosuccinimide-calcein reaction sensitized by zinc (II). | Zhao, F., et al. 2014. Luminescence. 29: 219-24. PMID: 23723137
- Immobilized N-chlorosuccinimide as a friendly peptide disulfide-forming reagent. | Postma, TM. and Albericio, F. 2014. ACS Comb Sci. 16: 160-3. PMID: 24641490
- N-chlorosuccinimide-promoted regioselective sulfenylation of imidazoheterocycles at room temperature. | Ravi, C., et al. 2014. Org Lett. 16: 2978-81. PMID: 24838116
- N-Chlorosuccinimide-mediated oxidative chlorination of thiols to Nα-protected amino alkyl sulfonyl azides and their utility in the synthesis of sulfonyl triazole acids. | Sharnabai, KM., et al. 2016. Protein Pept Lett.. PMID: 27908261
- Synthesis of Azobenzenes Using N-Chlorosuccinimide and 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU). | Antoine John, A. and Lin, Q. 2017. J Org Chem. 82: 9873-9876. PMID: 28846399
- N-Hydroxyphthalimide/benzoquinone-catalyzed chlorination of hydrocarbon C-H bond using N-chlorosuccinimide. | Li, ZH., et al. 2019. Org Biomol Chem. 17: 3403-3408. PMID: 30869109
- One-Pot Synthesis of P(O)-N Containing Compounds Using N-Chlorosuccinimide and Their Influence in Thermal Decomposition of PU Foams. | Salmeia, KA., et al. 2018. Polymers (Basel). 10: PMID: 30960665
- Visible-light-induced C(sp3)-H functionalizations of piperidines to 3,3-dichloro-2-hydroxy-piperidines with N-chlorosuccinimide. | Wang, F., et al. 2021. Org Biomol Chem. 19: 6141-6146. PMID: 34180488
- A Mechanistic Study for Aziridination of Nitroalkenes Mediated by N-Chlorosuccinimide. | Iwai, K., et al. 2022. J Oleo Sci. 71: 897-903. PMID: 35584955
- N-chlorosuccinimide enhancing the antimicrobial effect of benzalkonium chloride on biofilm Pseudomonas aeruginosa and its interaction mechanisms. | Huang, Z., et al. 2022. J Environ Sci Health A Tox Hazard Subst Environ Eng. 1-8. PMID: 35852222
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Chlorosuccinimide, 100 g | sc-250458 | 100 g | $24.00 |
