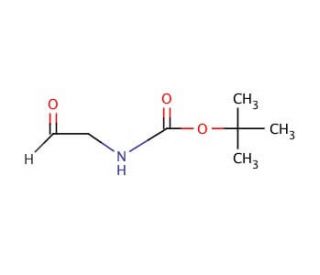
N-Boc-2-aminoacetaldehyde (CAS 89711-08-0)
QUICK LINKS
N-Boc-2-aminoacetaldehyde is a compound that functions as a versatile building block in organic synthesis. Its mechanism of action involves serving as a key intermediate in the preparation of various complex molecules, particularly those with agrochemical relevance. N-Boc-2-Aminoacetaldehyde undergoes specific chemical reactions, such as condensation and reduction, to form structurally diverse compounds with potential biological activity. In the development setting, N-Boc-2-aminoacetaldehyde is utilized for its ability to introduce specific functional groups and stereochemistry into target molecules, allowing for the exploration of structure-activity relationships and the development of novel chemical entities. Its role in the synthesis of diverse chemical scaffolds may be used for the creation of compound libraries and the discovery of new lead compounds for further investigation. At the molecular level, N-Boc-2-aminoacetaldehyde participates in various transformations, enabling the construction of complex molecular architectures with potential biological relevance.
N-Boc-2-aminoacetaldehyde (CAS 89711-08-0) References
- Modifications of the GSK3beta substrate sequence to produce substrate-mimetic inhibitors of Akt as potential anti-cancer therapeutics. | Kayser, KJ., et al. 2007. Bioorg Med Chem Lett. 17: 2068-73. PMID: 17276059
- Efficient total synthesis of (+)-negamycin, a potential chemotherapeutic agent for genetic diseases. | Hayashi, Y., et al. 2008. Chem Commun (Camb). 2379-81. PMID: 18473076
- Sense from nonsense: therapies for premature stop codon diseases. | Bidou, L., et al. 2012. Trends Mol Med. 18: 679-88. PMID: 23083810
- Tumor-targeting of EGFR inhibitors by hypoxia-mediated activation. | Karnthaler-Benbakka, C., et al. 2014. Angew Chem Int Ed Engl. 53: 12930-12935. PMID: 25079700
- Further insights into the SAR of α-substituted cyclopropylamine derivatives as inhibitors of histone demethylase KDM1A. | Pieroni, M., et al. 2015. Eur J Med Chem. 92: 377-86. PMID: 25585008
- Synthesis of triamino acid building blocks with different lipophilicities. | Maity, J., et al. 2015. PLoS One. 10: e0124046. PMID: 25876040
- Identification and SAR of Glycine Benzamides as Potent Agonists for the GPR139 Receptor. | Dvorak, CA., et al. 2015. ACS Med Chem Lett. 6: 1015-8. PMID: 26396690
- Enantioselective Synthesis of 3,3-Difluoropyrrolidin-4-ol, a Valuable Building Block in Medicinal Chemistry. | Si, C., et al. 2016. J Org Chem. 81: 4359-63. PMID: 27138111
- Discovery of N-(2-aminoethyl)-N-benzyloxyphenyl benzamides: New potent Trypanosoma brucei inhibitors. | Buchynskyy, A., et al. 2017. Bioorg Med Chem. 25: 1571-1584. PMID: 28187957
- Multicomponent Petasis Reaction for the Synthesis of Functionalized 2-Aminothiophenes and Thienodiazepines. | Hwang, J., et al. 2020. ACS Comb Sci. 22: 495-499. PMID: 32985878
- Anticancer properties of bisaminoquinolines with modified linkers. | Wang, Y., et al. 2021. Bioorg Med Chem Lett. 49: 128272. PMID: 34298133
- Efficacy and Selectivity of Monovalent and Bivalent Vaccination Strategies to Protect against Exposure to Carfentanil, Fentanyl, and Their Mixtures in Rats. | Crouse, B., et al. 2022. ACS Pharmacol Transl Sci. 5: 331-343. PMID: 35592436
- Implications of replacing peptide bonds in the COOH-terminal B chain domain of insulin by the psi (CH2-NH) linker. | Nakagawa, SH., et al. 1993. Int J Pept Protein Res. 42: 578-84. PMID: 8307689
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Boc-2-aminoacetaldehyde, 1 g | sc-257836 | 1 g | $99.00 |
