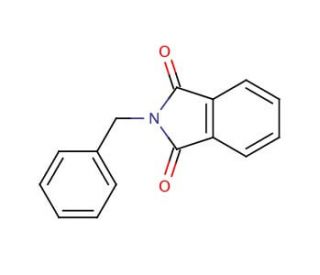
N-Benzylphthalimide (CAS 2142-01-0)
QUICK LINKS
N-Benzylphthalimide serves as a versatile reactive intermediate in various organic synthesis processes, underpinning the creation of a broad range of organic molecules used in non-medical applications. It is particularly valued for its efficacy in nucleophilic substitution reactions, a mechanism that facilitates the incorporation of the N-benzylphthalimide group into a variety of target molecules. This capability is for the synthesis of complex organic frameworks that are central to the production of advanced materials and agrochemicals. The compound′s ability to undergo selective functional group transformations is instrumental in constructing molecular scaffolds, for precise structural and stereochemical configurations.
N-Benzylphthalimide (CAS 2142-01-0) References
- Preparation of diazabicyclo[4.3.0]nonene-based peptidomimetics. | Hutton, CA. and Bartlett, PA. 2007. J Org Chem. 72: 6865-72. PMID: 17685573
- Kinetic coupled with UV spectral evidence for near-irreversible nonionic micellar binding of N-benzylphthalimide under the typical reaction conditions: an observation against a major assumption of the pseudophase micellar model. | Cheong, MY., et al. 2007. J Phys Chem B. 111: 12185-94. PMID: 17914797
- 2-Benzyl-isoindoline-1,3-dione: a monoclinic polymorph. | Jiang, Z., et al. 2007. Acta Crystallogr Sect E Struct Rep Online. 64: o324. PMID: 21200885
- The evaluation of new and isotopically labeled isoindoline nitroxides and an azaphenalene nitroxide for EPR oximetry. | Khan, N., et al. 2011. J Magn Reson. 211: 170-7. PMID: 21665499
- N-(4-Meth-oxy-benz-yl)phthalimide: a triclinic polymorph. | Takahashi, H. 2012. Acta Crystallogr Sect E Struct Rep Online. 68: o2457-8. PMID: 22904901
- Mechanochemical Lignin-Mediated Strecker Reaction. | Dabral, S., et al. 2017. Molecules. 22: PMID: 28106742
- Selective Hydrogenation of Cyclic Imides to Diols and Amines and Its Application in the Development of a Liquid Organic Hydrogen Carrier. | Kumar, A., et al. 2018. J Am Chem Soc. 140: 7453-7457. PMID: 29812921
- Paramagnetic Chemical Probes for Studying Biological Macromolecules. | Miao, Q., et al. 2022. Chem Rev. 122: 9571-9642. PMID: 35084831
- Spectroscopic determination of photostimulated reactions in halomethane solutions of phthalimide and its derivatives | V. V. Gurinovich, V. V. Gruzinskii & M. P. Tsvirko. (1988). Theoretical and Experimental Chemistry. volume 23,: pages450–454.
- 2,4,6-Trichloro-1,3,5-triazine/dimethylformamide as an efficient reagent for one-pot conversion of alcohols into N-alkylphthalimides | B Mokhtari, R Azadi, A Azhdari - Chinese Chemical Letters, 2010 - Elsevier. February 2010,. Chinese Chemical Letters. Volume 21, Issue 2,: Pages 171-174.
- Synthesis of 3-Benzylated Isoindolinones by Photoredox Decarboxylation of Arylacetates in the Presence of N-Benzylphthalimide: Conductivity as a Kinetic Tool | Prof. Dr. Axel G. Griesbeck, Dr. Jörg.-M. Neudörfl, Prof. Dr. Bernd Goldfuss, Dr. Sabrina Molitor. August 2017. ChemPhotoChem. Volume1, Issue8: Pages 355-362.
- Manganese catalyzed selective hydrogenation of cyclic imides to diols and amines†Check for updates | Uttam Kumar Das, ORCID logo a Trevor Janes,a Amit Kumar‡a and David Milstein ORCID logo *a. 2020,. Green Chem.,. 22,: 3079-3082.
- An Empirical Kinetic Approach To Study The Occurrence Of Ion‐Exchange At The Aqueous Cationic Micellar Surface: Alkaline Hydrolysis Of N‐Benzylphthalimide | Mohammad Niyaz Khan; May‐Ye Cheong; Azhar Ariffin. 1 June 2009. AIP Conference Proceedings. Volume 1136, Issue 1: 380–384.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Benzylphthalimide, 25 g | sc-236009 | 25 g | $109.00 |
