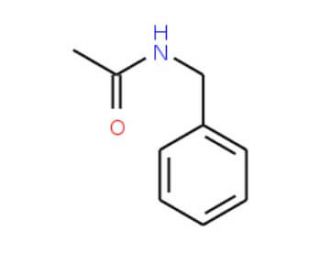
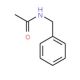
N-Benzylacetamide (CAS 588-46-5)
QUICK LINKS
N-Benzylacetamide is a compound that functions as an organic building block in chemical synthesis. N-Benzylacetamide undergoes reactions such as acylation, alkylation, and reduction to form more complex molecular structures. Its mechanism of action involves participating in organic reactions to introduce the N-benzylacetamide moiety into target molecules, leading to the formation of new chemical entities. N-Benzylacetamide′s role in chemical synthesis involves its ability to facilitate the creation of diverse chemical compounds through its reactivity and functional group compatibility. N-Benzylacetamide′s mechanism of action is characterized by its involvement in the formation of carbon-carbon and carbon-nitrogen bonds.
N-Benzylacetamide (CAS 588-46-5) References
- Phytoremediation potential of Portulaca grandiflora Hook. (Moss-Rose) in degrading a sulfonated diazo reactive dye Navy Blue HE2R (Reactive Blue 172). | Khandare, RV., et al. 2011. Bioresour Technol. 102: 6774-7. PMID: 21514149
- Automation of the CHARMM General Force Field (CGenFF) II: assignment of bonded parameters and partial atomic charges. | Vanommeslaeghe, K., et al. 2012. J Chem Inf Model. 52: 3155-68. PMID: 23145473
- A new synthetic route to N-benzyl carboxamides through the reverse reaction of N-substituted formamide deformylase. | Hashimoto, Y., et al. 2014. Appl Environ Microbiol. 80: 61-9. PMID: 24123742
- Identification of N-benzylacetamide as a major component of human plasmatic metabolic profiling of benznidazole. | Marson, ME., et al. 2015. Eur J Drug Metab Pharmacokinet. 40: 209-17. PMID: 24711214
- Antimicrobial Activity and Chromatographic Analysis of Extracts from Tropaeolum pentaphyllum Lam. Tubers. | da Cruz, RC., et al. 2016. Molecules. 21: PMID: 27136515
- Solvent-Free Iron(III) Chloride-Catalyzed Direct Amidation of Esters. | Mkhonazi, BD., et al. 2020. Molecules. 25: PMID: 32110915
- Discovery and Design of Family VIII Carboxylesterases as Highly Efficient Acyltransferases. | Müller, H., et al. 2021. Angew Chem Int Ed Engl. 60: 2013-2017. PMID: 33140887
- Catalytic Oxidative Deamination by Water with H2 Liberation. | Tang, S., et al. 2020. J Am Chem Soc. 142: 20875-20882. PMID: 33237749
- Tandem mass spectrometry of aqueous extract from Ficus dubia sap and its cell-based assessments for use as a skin antioxidant. | Chansriniyom, C., et al. 2021. Sci Rep. 11: 16899. PMID: 34413383
- Research Progress and Key Issues of Hydrodebenzylation of Hexabenzylhexaazaisowurtzitane (HBIW) in the Synthesis of High Energy Density Material Hexanitrohexaazaisowurtzitane (HNIW). | Tang, X., et al. 2022. Materials (Basel). 15: PMID: 35057135
- Cobalt catalysed aminocarbonylation of thiols in batch and flow for the preparation of amides. | Orduña, JM., et al. 2021. RSC Adv. 11: 30398-30406. PMID: 35480268
- Controllable Assembly of Vanadium-Containing Polyoxoniobate-Based Materials and Their Electrocatalytic Activity for Selective Benzyl Alcohol Oxidation. | Li, X., et al. 2022. Molecules. 27: PMID: 35566213
- An extensive review of marine pigments: sources, biotechnological applications, and sustainability. | Muñoz-Miranda, LA. and Iñiguez-Moreno, M. 2023. Aquat Sci. 85: 68. PMID: 37096011
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Benzylacetamide, 1 g | sc-295667B | 1 g | $32.00 | |||
N-Benzylacetamide, 5 g | sc-295667 | 5 g | $52.00 | |||
N-Benzylacetamide, 25 g | sc-295667A | 25 g | $188.00 | |||
N-Benzylacetamide, 100 g | sc-295667C | 100 g | $530.00 |
