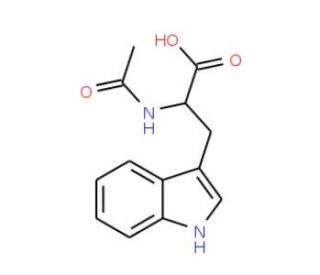
Molecular structure of N-Acetyl-D-tryptophan, CAS Number: 2280-01-5
N-Acetyl-D-tryptophan (CAS 2280-01-5)
CAS Number:
2280-01-5
Molecular Weight:
246.26
Molecular Formula:
C13H14N2O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
N-Acetyl-D-tryptophan is a compound that functions as a precursor in the synthesis of proteins and neurotransmitters. At the molecular level, N-Acetyl-D-tryptophan is involved in the biosynthesis of serotonin, where it is converted into 5-hydroxytryptophan by the enzyme tryptophan hydroxylase. N-Acetyl-D-tryptophan′s mechanism of action involves its incorporation into proteins and its conversion into serotonin, contributing to the regulation of neurotransmission and protein synthesis. Its function in these processes may be an important compound for understanding the molecular mechanisms underlying neurotransmitter regulation and protein synthesis in experimental applications.
N-Acetyl-D-tryptophan (CAS 2280-01-5) References
- Identification and characterization of a new gene from Variovorax paradoxus Iso1 encoding N-acyl-D-amino acid amidohydrolase responsible for D-amino acid production. | Lin, PH., et al. 2002. Eur J Biochem. 269: 4868-78. PMID: 12354118
- Studies on acylase activity and micro-organisms. XX. Isolation of soil bacteria capable of resolving trytophan by their acylase on N-acetyl-DL-tryptophan. | KAMEDA, Y. and ISHII, Y. 1962. Chem Pharm Bull (Tokyo). 10: 1152-3. PMID: 13962055
- Studies on acylase activity and micro-organisms. XIX. Optical resolution of threonine. lvsine. trvptophan. and methionine by ucylase of soil bacteria. | KAMEDA, Y., et al. 1962. Chem Pharm Bull (Tokyo). 10: 1146-51. PMID: 13962056
- D-amino acid N-acetyltransferase of Saccharomyces cerevisiae: a close homologue of histone acetyltransferase Hpa2p acting exclusively on free D-amino acids. | Yow, GY., et al. 2004. Arch Microbiol. 182: 396-403. PMID: 15375647
- Purification and Characterization of d-Aminoacylase from Alcaligenes faecalis DA1. | Yang, YB., et al. 1991. Appl Environ Microbiol. 57: 1259-60. PMID: 16348465
- Induction of the d-Amino Acid Oxidase from Trigonopsis variabilis. | Horner, R., et al. 1996. Appl Environ Microbiol. 62: 2106-10. PMID: 16535339
- Annotating enzymes of uncertain function: the deacylation of D-amino acids by members of the amidohydrolase superfamily. | Cummings, JA., et al. 2009. Biochemistry. 48: 6469-81. PMID: 19518059
- N-acetyl-l-tryptophan, but not N-acetyl-d-tryptophan, rescues neuronal cell death in models of amyotrophic lateral sclerosis. | Sirianni, AC., et al. 2015. J Neurochem. 134: 956-68. PMID: 26031348
- Exploratory metabolomics of nascent metabolic syndrome. | Shim, K., et al. 2019. J Diabetes Complications. 33: 212-216. PMID: 30611573
- Complete Stereoinversion of l-Tryptophan by a Fungal Single-Module Nonribosomal Peptide Synthetase. | Hai, Y., et al. 2019. J Am Chem Soc. 141: 16222-16226. PMID: 31573806
- Correcting a widespread error: Neuroprotectant N-acetyl-L-tryptophan does not bind to the neurokinin-1 receptor. | Matalińska, J. and Lipiński, PFJ. 2022. Mol Cell Neurosci. 120: 103728. PMID: 35421568
- Significant metabolic alterations in non-small cell lung cancer patients by epidermal growth factor receptor-targeted therapy and PD-1/PD-L1 immunotherapy. | Yan, C., et al. 2022. Front Pharmacol. 13: 949745. PMID: 36034789
- Calorimetric investigations of the binding of inhibitors to alpha-chymotrypsin. I. The enthalpy of dilution of alpha-chymotrypsin and of proflavin, and the enthalpy of binding of indole, N-acetyl-D-tryptophan, and proflavin to alpha-chymotrypsin. | Shiao, DD. and Sturtevant, JM. 1969. Biochemistry. 8: 4910-7. PMID: 5391863
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Acetyl-D-tryptophan, 1 g | sc-301270 | 1 g | $24.00 | |||
N-Acetyl-D-tryptophan, 5 g | sc-301270A | 5 g | $52.00 |
