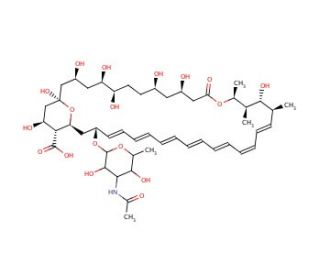
N-Acetyl Amphotericin B (CAS 902457-23-2)
QUICK LINKS
N-Acetyl Amphotericin B, a derivative of the antifungal agent amphotericin B, has garnered attention in scientific research for its potential applications in understanding the structure-activity relationships of amphotericin B and exploring novel antifungal strategies. This compound is synthesized through the acetylation of amphotericin B, resulting in modifications to its chemical structure while preserving its antifungal properties. Research has focused on elucidating the mechanisms of action of N-Acetyl Amphotericin B, which are believed to be similar to those of amphotericin B. Specifically, both compounds are thought to exert their antifungal effects by binding to ergosterol, a key component of fungal cell membranes, and forming pores that disrupt membrane integrity, leading to leakage of cellular contents and ultimately fungal cell death. Furthermore, studies have investigated the efficacy of N-Acetyl Amphotericin B against various fungal pathogens, including multidrug-resistant strains, highlighting its potential as a lead compound for the development of novel antifungal agents. Additionally, N-Acetyl Amphotericin B has served as a valuable tool in structure-activity relationship studies, aiding in the design and optimization of more potent and less toxic antifungal compounds. Overall, N-Acetyl Amphotericin B represents a promising avenue for further exploration in antifungal drug discovery and as a tool for investigating fungal membrane biology.
N-Acetyl Amphotericin B (CAS 902457-23-2) References
- Single-dose AmBisome (Liposomal amphotericin B) as prophylaxis for murine systemic candidiasis and histoplasmosis. | Garcia, A., et al. 2000. Antimicrob Agents Chemother. 44: 2327-32. PMID: 10952575
- Determination of amphotericin B in human plasma using solid-phase extraction and high-performance liquid chromatography. | Eldem, T. and Arican-Cellat, N. 2001. J Pharm Biomed Anal. 25: 53-64. PMID: 11274858
- Comparative efficacies, toxicities, and tissue concentrations of amphotericin B lipid formulations in a murine pulmonary aspergillosis model. | Olson, JA., et al. 2006. Antimicrob Agents Chemother. 50: 2122-31. PMID: 16723574
- Sensitive high-pressure liquid chromatographic assay for amphotericin B which incorporates an internal standard. | Granich, GG., et al. 1986. Antimicrob Agents Chemother. 29: 584-8. PMID: 3707107
- Physico-chemical and microbiological comparison of nystatin, amphotericin A and amphotericin B, and structure of amphotericin A. | Aszalos, A., et al. 1985. J Antibiot (Tokyo). 38: 1699-713. PMID: 3912360
- Molecular aspects of polyene- and sterol-dependent pore formation in thin lipid membranes. | Dennis, VW., et al. 1970. J Gen Physiol. 55: 375-400. PMID: 4938534
- Prophylactic efficacy of aerosolized liposomal (AmBisome) and non-liposomal (Fungizone) amphotericin B in murine pulmonary aspergillosis. | Allen, SD., et al. 1994. J Antimicrob Chemother. 34: 1001-13. PMID: 7730214
- Determination of amphotericin B in cerebrospinal fluid by solid-phase extraction and liquid chromatography. | Liu, H., et al. 1995. J Pharm Biomed Anal. 13: 1395-400. PMID: 8634357
- Enhanced encapsulation of amphotericin B into liposomes by complex formation with polyethylene glycol derivatives. | Moribe, K., et al. 1998. Pharm Res. 15: 1737-42. PMID: 9833996
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Acetyl Amphotericin B, 2.5 mg | sc-212073 | 2.5 mg | $284.00 | |||
N-Acetyl Amphotericin B, 25 mg | sc-212073A | 25 mg | $2290.00 | |||
N-Acetyl Amphotericin B, 50 mg | sc-212073B | 50 mg | $3000.00 |
