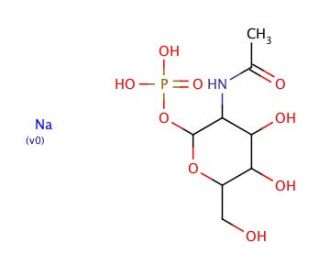
Molecular structure of N-Acetyl-α-D-glucosamine 1-phosphate disodium salt, CAS Number: 31281-59-1
N-Acetyl-α-D-glucosamine 1-phosphate disodium salt (CAS 31281-59-1)
Alternate Names:
n-acetyl-α-d-glucosamine 1-phosphate disodium salt
Application:
N-Acetyl-α-D-glucosamine 1-phosphate disodium salt is a substrate used to identify, differentiate and characterize N-acetyl-D-glucosamine-phosphate deacetylases
CAS Number:
31281-59-1
Purity:
≥95%
Molecular Weight:
345.15
Molecular Formula:
C8H16NO9P•2Na
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
N-Acetyl-α-D-glucosamine 1-phosphate disodium salt is a substrate used to identify, differentiate and characterize N-acetyl-D-glucosamine-phosphate deacetylases. This compound demonstrates a diverse array of biochemical and physiological effects. Extensive studies have revealed its efficacy against an array of ailments encompassing cancer, inflammation, and neurological disorders. The underlying mechanism of action is thought to involve the inhibition of critical enzymes and signaling pathways.
N-Acetyl-α-D-glucosamine 1-phosphate disodium salt (CAS 31281-59-1) References
- Oxidation of 3-C-(2-amino-2-deoxy-D-glucopyranosyl)-1-propene compounds and the structure of 3-C-(2-amino-2-deoxy-D-glucopyranosyl)-1,2-propanediol derivatives for a synthesis of 2,3-didehydro-2,7-dideoxy-N-acetylneuraminic acid. | Machinami, T., et al. 2002. Carbohydr Res. 337: 1917-27. PMID: 12433457
- Pathological consequences of VCP mutations on human striated muscle. | Hübbers, CU., et al. 2007. Brain. 130: 381-93. PMID: 16984901
- A convenient synthesis of the C-1-phosphonate analogue of UDP-GlcNAc and its evaluation as an inhibitor of O-linked GlcNAc transferase (OGT). | Hajduch, J., et al. 2008. Carbohydr Res. 343: 189-95. PMID: 18039537
- Exploring specificity of glycosyltransferases: synthesis of new sugar nucleotide related molecules as putative donor substrates. | Khaled, A., et al. 2008. Carbohydr Res. 343: 167-78. PMID: 18048019
- The first C-glycosidic analogue of a novel galactosyltransferase inhibitor. | Descroix, K. and Wagner, GK. 2011. Org Biomol Chem. 9: 1855-63. PMID: 21267505
- Characterization of size, structure and purity of serogroup X Neisseria meningitidis polysaccharide, and development of an assay for quantification of human antibodies. | Xie, O., et al. 2012. Vaccine. 30: 5812-23. PMID: 22835740
- Synthesis and evaluation of l-rhamnose 1C-phosphonates as nucleotidylyltransferase inhibitors. | Loranger, MW., et al. 2013. J Org Chem. 78: 9822-33. PMID: 24020932
- Evidence of a new intermediate compound of the chitin biogenesis found in a marine-derived fungus. | Toledo Marante, FJ., et al. 2014. Appl Biochem Biotechnol. 174: 2426-34. PMID: 25205173
- Effect of the urease-derived peptide Jaburetox on the central nervous system of Triatoma infestans (Insecta: Heteroptera). | Galvani, GL., et al. 2015. Biochim Biophys Acta. 1850: 255-62. PMID: 25445710
- A kinetic study of the effect of alpha-D-galactose, alpha-D-mannose, alpha-D-glucosamine, N-acetyl-alpha-D-glucosamine, and alpha-D-ribose diphosphate on the activity of phosphoglucomutase. | Mulhausen, H. and Mendicino, J. 1970. J Biol Chem. 245: 4038-46. PMID: 4395681
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Acetyl-α-D-glucosamine 1-phosphate disodium salt, 5 mg | sc-215434 | 5 mg | $430.00 | |||
N-Acetyl-α-D-glucosamine 1-phosphate disodium salt, 25 mg | sc-215434A | 25 mg | $960.00 |
