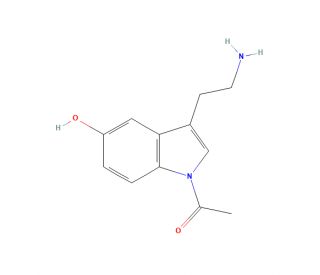
N-Acetyl-5-hydroxytryptamine (CAS 1210-83-9)
QUICK LINKS
N-Acetyl-5-hydroxytryptamine functions as a neurotransmitter and hormone in various organisms. It acts as a modulator of physiological processes, including circadian rhythms, mood, and behavior. At the molecular level, it interacts with specific receptors in the central nervous system, particularly the serotonin receptors, to regulate neurotransmission and signal transduction. N-Acetyl-5-Hydroxytryptamine also plays a role in the regulation of sleep, appetite, and stress responses. N-Acetyl-5-hydroxytryptamine is involved in the modulation of gastrointestinal function and cardiovascular activity. Its mechanism of action involves binding to specific target sites and influencing intracellular signaling pathways, ultimately impacting various physiological functions.
N-Acetyl-5-hydroxytryptamine (CAS 1210-83-9) References
- Melatonin and its precursors scavenge nitric oxide. | Noda, Y., et al. 1999. J Pineal Res. 27: 159-63. PMID: 10535765
- Chemiluminescence determination of melatonin and some of its derivatives using potassium permanganate and formaldehyde system. | Chen, GN., et al. 2003. Anal Bioanal Chem. 376: 873-8. PMID: 12827333
- The metabolism of serotonin (5-hydroxytryptamine). | McISAAC, WM. and PAGE, IH. 1959. J Biol Chem. 234: 858-64. PMID: 13654278
- Reaction mechanism of melatonin oxidation by reactive oxygen species in vitro. | Bonnefont-Rousselot, D., et al. 2011. J Pineal Res. 50: 328-35. PMID: 21244479
- Selective and sensitive liquid chromatographic determination method of 5-hydroxyindoles with fluorous and fluorogenic derivatization. | Sakaguchi, Y., et al. 2015. J Pharm Biomed Anal. 114: 348-54. PMID: 26112924
- Quercetin Suppresses AOM/DSS-Induced Colon Carcinogenesis through Its Anti-Inflammation Effects in Mice. | Lin, R., et al. 2020. J Immunol Res. 2020: 9242601. PMID: 32537472
- Construction of cell factory capable of efficiently converting L-tryptophan into 5-hydroxytryptamine. | Wang, Y., et al. 2022. Microb Cell Fact. 21: 47. PMID: 35331215
- Circ-ERC2 Is Involved in Melatonin Synthesis by Regulating the miR-125a-5p/MAT2A Axis. | Guo, HX., et al. 2022. Int J Mol Sci. 23: PMID: 36555116
- Hydroxyindole-O-methyltransferase. II. Inhibitory activities of some N-acyltryptamines. | Ho, BT., et al. 1968. J Pharm Sci. 57: 1998-2000. PMID: 5725938
- Hydroxyindole-O-methyltransferase. I. Substrate binding. | Ho, BT., et al. 1969. J Pharm Sci. 58: 130-1. PMID: 5765823
- Distribution of 5-hydroxytryptamine and indolealkylamine metabolites in the American cockroach, Periplaneta americana L. | Sloley, BD. and Downer, RG. 1984. Comp Biochem Physiol C Comp Pharmacol Toxicol. 79: 281-6. PMID: 6083843
- Inhibition by N-acetyl-5-hydroxytryptamine of nitric oxide synthase expression in cultured cells and in the anaesthetized rat. | Klemm, P., et al. 1995. Br J Pharmacol. 115: 1175-81. PMID: 7582541
- The determination of 5-hydroxytryptamine, related indolealkylamines and 5-hydroxyindoleacetic acid in the bovine eye by gas chromatography-negative ion chemical ionization mass spectrometry. | Best, SA., et al. 1993. J Pharm Biomed Anal. 11: 323-33. PMID: 7689343
- gamma-Glutamyl conjugation of 5-hydroxytryptamine (serotonin) in the earthworm (Lumbricus terrestris). | Sloley, BD. 1994. Neurochem Res. 19: 217-22. PMID: 7910382
- In-vivo microdialysis measurement of 5-hydroxytryptamine and its metabolites, 5-hydroxyindoleacetic acid and N-acetyl 5-hydroxytryptamine, in rat blood: effects of histamine-receptor antagonists. | Sakurai, E., et al. 1996. J Pharm Pharmacol. 48: 911-3. PMID: 8910852
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Acetyl-5-hydroxytryptamine, 10 mg | sc-202716 | 10 mg | $20.00 | |||
N-Acetyl-5-hydroxytryptamine, 50 mg | sc-202716A | 50 mg | $104.00 |
