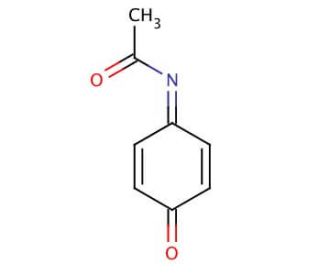
N-Acetyl-4-benzoquinone Imine (CAS 50700-49-7)
See product citations (2)
QUICK LINKS
N-Acetyl-4-benzoquinone Imine, or Acetimidoquinone (AIQ), is a toxic metabolite of Acetaminophen, which is known to react with serum proteins. It functions as an antioxidant, a chelating agent, and an inhibitor of enzymes, such as cytochrome P450 and DNA polymerase. On the other hand, it may act to increase the activity of detoxifying enzymes, such as glutathione S-transferase. N-Acetyl-4-benzoquinone Imine finds many applications in scientific research. For example, it is used in the synthesis of antibiotics, antifungal agents, anti-inflammatory agents, dyes, agrochemicals, proteins, enzymes, and hormones. It is also employed in the study of the structure and function of proteins, enzymes, and hormones.
N-Acetyl-4-benzoquinone Imine (CAS 50700-49-7) References
- Causes of Alzheimer's disease: paracetamol (acetaminophen) today? Amphetamines tomorrow? | Jones, GR. 2001. Med Hypotheses. 56: 121-3. PMID: 11133268
- The role of oxidant stress and reactive nitrogen species in acetaminophen hepatotoxicity. | Jaeschke, H., et al. 2003. Toxicol Lett. 144: 279-88. PMID: 12927346
- N-acetyl-p-benzoquinone imine-induced protein thiol modification in isolated rat hepatocytes. | Weis, M., et al. 1992. Biochem Pharmacol. 43: 1493-505. PMID: 1567474
- Intracellular signaling mechanisms of acetaminophen-induced liver cell death. | Jaeschke, H. and Bajt, ML. 2006. Toxicol Sci. 89: 31-41. PMID: 16177235
- Transformation of acetaminophen by chlorination produces the toxicants 1,4-benzoquinone and N-acetyl-p-benzoquinone imine. | Bedner, M. and MacCrehan, WA. 2006. Environ Sci Technol. 40: 516-22. PMID: 16468397
- On the chemistry of the reaction between N-acetylcysteine and 4-[(4-ethoxyphenyl)imino]-2,5-cyclohexadien-1-one, a 4-ethoxyaniline metabolite formed during peroxidase reactions. | Lindqvist, T., et al. 1991. Chem Res Toxicol. 4: 489-96. PMID: 1912338
- Early loss of large genomic DNA in vivo with accumulation of Ca2+ in the nucleus during acetaminophen-induced liver injury. | Ray, SD., et al. 1990. Toxicol Appl Pharmacol. 106: 346-51. PMID: 2256122
- Glutathione disulfide formation and oxidant stress during acetaminophen-induced hepatotoxicity in mice in vivo: the protective effect of allopurinol. | Jaeschke, H. 1990. J Pharmacol Exp Ther. 255: 935-41. PMID: 2262912
- Integrated digital microfluidic platform for voltammetric analysis. | Dryden, MD., et al. 2013. Anal Chem. 85: 8809-16. PMID: 24001207
- Formation of 4-aminophenoxyl free radical from the acetaminophen metabolite N-acetyl-p-benzoquinone imine. | Fischer, V., et al. 1985. J Biol Chem. 260: 11446-50. PMID: 2995335
- Comparative cytotoxic effects of N-acetyl-p-benzoquinone imine and two dimethylated analogues. | Rundgren, M., et al. 1988. Mol Pharmacol. 34: 566-72. PMID: 3173335
- PiTMaP: A New Analytical Platform for High-Throughput Direct Metabolome Analysis by Probe Electrospray Ionization/Tandem Mass Spectrometry Using an R Software-Based Data Pipeline. | Zaitsu, K., et al. 2020. Anal Chem. 92: 8514-8522. PMID: 32375466
- Metal-organic framework precursors derived Ni-doping porous carbon spheres for sensitive electrochemical detection of acetaminophen. | Guo, L., et al. 2021. Talanta. 228: 122228. PMID: 33773732
- Antioxidant and Prooxidant Effects of Thymoquinone and Its Hydroquinone Metabolite. | Mizuno, M. and Fukuhara, K. 2022. Biol Pharm Bull. 45: 1389-1393. PMID: 36047210
- Reactions of N-acetyl-p-benzoquinone imine with reduced glutathione, acetaminophen, and NADPH. | Potter, DW. and Hinson, JA. 1986. Mol Pharmacol. 30: 33-41. PMID: 3724743
- Detection of N-acetyl-p-benzoquinone imine produced during the hydrolysis of the model phenacetin metabolite N-(pivaloyloxy)phenacetin. | Novak, M., et al. 1986. J Med Chem. 29: 1424-9. PMID: 3735311
- Mechanisms of acetaminophen oxidation to N-acetyl-P-benzoquinone imine by horseradish peroxidase and cytochrome P-450. | Potter, DW. and Hinson, JA. 1987. J Biol Chem. 262: 966-73. PMID: 3805031
- Mechanisms of N-acetyl-p-benzoquinone imine cytotoxicity. | Albano, E., et al. 1985. Mol Pharmacol. 28: 306-11. PMID: 4033631
- Reduction and glutathione conjugation reactions of N-acetyl-p-benzoquinone imine and two dimethylated analogues. | Rosen, GM., et al. 1984. Mol Pharmacol. 25: 151-7. PMID: 6323948
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Acetyl-4-benzoquinone Imine, 1 mg | sc-212083 | 1 mg | $243.00 | |||
N-Acetyl-4-benzoquinone Imine, 2 mg | sc-212083A | 2 mg | $426.00 | |||
N-Acetyl-4-benzoquinone Imine, 5 mg | sc-212083B | 5 mg | $634.00 | |||
N-Acetyl-4-benzoquinone Imine, 10 mg | sc-212083C | 10 mg | $1093.00 | |||
N-Acetyl-4-benzoquinone Imine, 25 mg | sc-212083D | 25 mg | $1987.00 |
