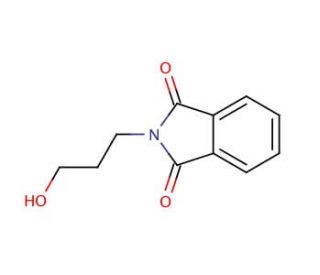
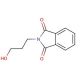
N-(3-Hydroxypropyl)phthalimide (CAS 883-44-3)
QUICK LINKS
N-(3-Hydroxypropyl)phthalimide (NHP) is a synthetic compound that finds diverse applications in scientific research. Derived from phthalimide, a heterocyclic compound containing nitrogen, N-(3-Hydroxypropyl)phthalimid serves as a proton-acceptor in acid-base reactions. Its utility extends to acting as a coupling reagent in the synthesis of various compounds, including peptides and peptidomimetics. Moreover, N-(3-Hydroxypropyl)phthalimid proves valuable in the synthesis of organometallic compounds and contributes to the formation of metal-organic frameworks. Within scientific research, N-(3-Hydroxypropyl)phthalimide has proven versatile. It serves as a coupling reagent, facilitating the synthesis of peptides and peptidomimetics, while also participating in the formation of metal-organic frameworks. Furthermore, it acts as a proton-acceptor during acid-base reactions and plays a role as a reagent in the synthesis of organometallic compounds. The mechanism of action for N-(3-Hydroxypropyl)phthalimide lies in its ability to function as a proton-acceptor in acid-base reactions. In these reactions, N-(3-Hydroxypropyl)phthalimid acts as a nucleophile, accepting a proton from an acid and forming a new bond with it. This fundamental reaction plays a role in the creation of organometallic compounds, as well as in the synthesis of peptides and peptidomimetics.
N-(3-Hydroxypropyl)phthalimide (CAS 883-44-3) References
- Parallel synthesis and biological activity of a new class of high affinity and selective delta-opioid ligand. | Barn, DR., et al. 2001. Bioorg Med Chem. 9: 2609-24. PMID: 11557349
- Synthesis and characterization of PNA oligomers containing preQ1 as a positively charged guanine analogue. | Moriya, SS., et al. 2021. Bioorg Med Chem Lett. 39: 127850. PMID: 33662538
- Metabolic profiling in tissues and urine of patients with prostatic lesions and the diagnostic value of urine extracellular vesicles metabolites in prostate cancer. | Ding, T., et al. 2024. Clin Chim Acta. 556: 117845. PMID: 38403146
- Rapid and clean synthesis of phthalimide derivatives in high-temperature, high-pressure H2O/EtOH mixtures | Joan Fraga-Dubreuil, Gürbüz Çomak, Alasdair W. Taylora and Martyn Poliakoff. 2007. Green Chem. 9: 1067-1072.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-(3-Hydroxypropyl)phthalimide, 25 g | sc-228634 | 25 g | $51.00 |
