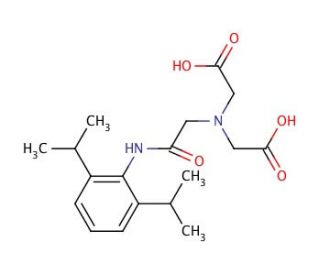
N-(2,6-Diisopropylphenylcarbamoylmethyl)iminodiacetic Acid (CAS 65717-97-7)
QUICK LINKS
N-(2,6-Diisopropylphenylcarbamoylmethyl)iminodiacetic Acid is a specialized chemical agent extensively utilized in the field of coordination chemistry, particularly in the synthesis and stabilization of metal complexes. This compound features a carbamoylmethyl group linked to iminodiacetic acid, which plays a critical role in chelating metal ions, thus stabilizing them in a solution. Its distinctive structure, incorporating diisopropylphenyl groups, enhances its lipophilicity and modifies the electronic environment of the metal center, influencing the reactivity and stability of the complex. This attribute is particularly valuable in catalysis research, where the controlled reactivity of metal complexes is crucial. Researchers leverage this compound to investigate the mechanistic pathways of catalyzed reactions, such as oxidation or polymerization processes, by observing how the changes in metal coordination affect the reaction outcomes. Additionally, its use extends to the design of novel organometallic frameworks, where it facilitates the formation of architectures with specific electronic properties for use in materials science, especially in the development of sensors and conductive materials. The ability to fine-tune the electronic characteristics of metal centers via such ligands is fundamental in advancing the understanding of metal-ligand interactions and their applications in various scientific domains.
N-(2,6-Diisopropylphenylcarbamoylmethyl)iminodiacetic Acid (CAS 65717-97-7) References
- Determining Amount of Hydrolyzed-Reduced Technetium: Alternate Method. | Mayer, Wilhelm J. 1982. Journal of Nuclear Medicine Technology. 10.4: 229-229.
- Gadolinium labeled pharmaceuticals as potential mri contrast agents for liver and biliary tract. | Najafi, A., et al. 1987. Journal of Labelled Compounds and Radiopharmaceuticals. 24.9: 1131-1141.
- A study of the preparation of disofenin and gluceptate cold-unit doses. | Cheng, Kenneth T., et al. 1992. Journal of nuclear medicine technology. 20.1: 33-35.
- Synthesis and characterization of GaIII, YIII, and LuIII complexes with etifenin and analogues. | Pfeifer‐Leeg and Michel, et al. 2016. Zeitschrift für anorganische und allgemeine Chemie. 642.6: 486-491.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-(2,6-Diisopropylphenylcarbamoylmethyl)iminodiacetic Acid, 1 g | sc-211962 | 1 g | $320.00 |
