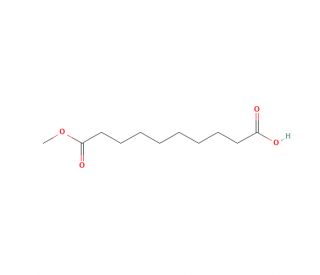
Monomethyl Sebacate (CAS 818-88-2)
QUICK LINKS
Monomethyl Sebacate is a chemical compound that is primarily known for its role as an intermediate in the synthesis of various industrial products. It is a monoester derived from sebacic acid and methanol. In the realm of polymer science, Monomethyl Sebacate is used as a reactant to produce polyesters and as a plasticizer in the manufacture of plastics, imparting flexibility and durability to the final products. It serves as an important compound in the synthesis of biodegradable polymers, which are of significant interest due to their environmental benefits. Additionally, Monomethyl Sebacate is utilized in the study of organic synthesis processes, providing insight into esterification reactions and the optimization of catalysts used in such chemical transformations.
Monomethyl Sebacate (CAS 818-88-2) References
- Contributions of flumequine and nitroarenes to the genotoxicity of river and ground waters. | Ma, F., et al. 2012. Chemosphere. 88: 476-83. PMID: 22483724
- Synthesis and structure-activity relationship of non-phosphorus-based fructose-1,6-bisphosphatase inhibitors: 2,5-Diphenyl-1,3,4-oxadiazoles. | Liao, BR., et al. 2014. Eur J Med Chem. 83: 15-25. PMID: 24946215
- N-alkyl-hydroxybenzoyl anilide hydroxamates as dual inhibitors of HDAC and HSP90, downregulating IFN-γ induced PD-L1 expression. | Mehndiratta, S., et al. 2020. Eur J Med Chem. 185: 111725. PMID: 31655430
- Systematically characterized mechanism of treatment for lumbar disc herniation based on Yaobitong capsule ingredient analysis in rat plasma and its network pharmacology strategy by UPLC-MS/MS. | Deng, Y., et al. 2020. J Ethnopharmacol. 260: 113097. PMID: 32531413
- A Comparison of the Antibacterial Efficacy of Carbohydrate Lipid-like (Thio)Ether, Sulfone, and Ester Derivatives against Paenibacillus larvae. | Šamšulová, V., et al. 2023. Molecules. 28: PMID: 36985490
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Monomethyl Sebacate, 5 g | sc-474435 | 5 g | $202.00 |
