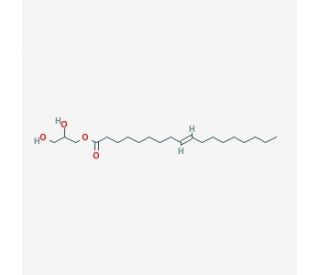
Monoelaidin (CAS 2716-53-2)
QUICK LINKS
Monoelaidin is a biocompatible polymer derived from monoolein. Monoelaidin possesses a phase transition temperature of approximately 37°C. Further, it demonstrates a strong affinity for adsorbing sodium ions and other cations, making it highly suitable for ion-exchange purposes. Notably, this polymer exhibits complete solubility in water or glycerol and has the capability to form bilayers. These bilayers are essential lipid membranes that play a critical role in cell structure and function. Monoelaidin synthesis involves the reaction between oleic acid and ethylene oxide and propylene oxide, which results in the ester linkage between two molecules of propylene glycol. During this process, disulfide bonds are formed, leading to the creation of crystalline phases that can serve as experimental models for studying disulfide bonds.
Monoelaidin (CAS 2716-53-2) References
- Monoolein: a review of the pharmaceutical applications. | Ganem-Quintanar, A., et al. 2000. Drug Dev Ind Pharm. 26: 809-20. PMID: 10900537
- Self-assembly in monoelaidin aqueous dispersions: direct vesicles to cubosomes transition. | Yaghmur, A., et al. 2008. PLoS One. 3: e3747. PMID: 19015726
- Nanostructural studies on monoelaidin-water systems at low temperatures. | Kulkarni, CV. 2011. Langmuir. 27: 11790-800. PMID: 21846133
- Self-assembled nanostructures of fully hydrated monoelaidin-elaidic acid and monoelaidin-oleic acid systems. | Yaghmur, A., et al. 2012. Langmuir. 28: 10105-19. PMID: 22690845
- Lipid-based Nanocarriers for siRNA Delivery: Challenges, Strategies and the Lessons Learned from the DODAX: MO Liposomal System. | Oliveira, ACN., et al. 2019. Curr Drug Targets. 20: 29-50. PMID: 29968536
- Protein-Eye View of the in Meso Crystallization Mechanism. | van T Hag, L., et al. 2019. Langmuir. 35: 8344-8356. PMID: 31122018
- Recombinant human MMP-2 associated with monoolein improves bone repair. | de Figueiredo, FAT., et al. 2020. Bratisl Lek Listy. 121: 571-579. PMID: 32726120
- Gelatin-loaded cubosomes stabilized with hydrophobically modified quaternized cellulose nanofiber and their pH-dependent release property. | Park, SH., et al. 2021. J Biomater Appl. 35: 1109-1118. PMID: 33632005
- Kinetics and mechanism of transitions involving the lamellar, cubic, inverted hexagonal, and fluid isotropic phases of hydrated monoacylglycerides monitored by time-resolved X-ray diffraction. | Caffrey, M. 1987. Biochemistry. 26: 6349-63. PMID: 3427010
- Impact of macromolecular crowding on the mesomorphic behavior of lipid self-assemblies. | Mangiarotti, A. and Bagatolli, LA. 2021. Biochim Biophys Acta Biomembr. 1863: 183728. PMID: 34416246
- Tuning curvature and phase behavior of monoolein bilayers by epigallocatechin-3-gallate: Structural insight and cytotoxicity. | Minnelli, C., et al. 2022. Colloids Surf B Biointerfaces. 209: 112171. PMID: 34736221
- Antioxidant-containing monoolein aqueous dispersions: a preliminary study. | Sguizzato, M., et al. 2022. Drug Deliv Transl Res. 12: 1873-1880. PMID: 35084708
- Structure-rheology relationship in monoolein liquid crystals. | Mistry, S., et al. 2023. J Colloid Interface Sci. 630: 878-887. PMID: 36356453
- Characterizing the Self-Assembly Properties of Monoolein Lipid Isosteres. | Fracassi, A., et al. 2023. J Phys Chem B. 127: 1771-1779. PMID: 36795462
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Monoelaidin, 1 g | sc-295493 | 1 g | $167.00 |
